Testosterone (medication)
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | tess-toss-ter-own[1] |
| Trade names | Androgel, Testim, Testogel, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a614041 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Transdermal (gel, cream, solution, patch), by mouth (as testosterone undecanoate), in the cheek, intranasal (gel), intramuscular injection (as esters), subcutaneous pellets |
| ATC code | G03BA03 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Oral: very low (due to extensive first pass metabolism) |
| Protein binding | 97.0–99.5% (to SHBG and albumin)[2] |
| Metabolism | Liver (mainly reduction and conjugation) |
| Biological half-life | 2–4 hours |
| Excretion | Urine (90%), feces (6%) |
| Identifiers | |
| |
| Synonyms | Androst-4-en-17β-ol-3-one |
| CAS Number |
58-22-0 57-85-2 (propionate) 315-37-7 (enanthate) 58-20-8 (cypionate) 5949-44-0 (undecanoate) |
| PubChem (CID) | 6013 |
| IUPHAR/BPS | 2858 |
| DrugBank |
DB00624 |
| ChemSpider |
5791 |
| UNII |
3XMK78S47O |
| KEGG |
D00075 |
| ChEBI |
CHEBI:17347 |
| ChEMBL |
CHEMBL386630 |
| Chemical and physical data | |
| Formula | C19H28O2 |
| Molar mass | 288.42 g/mol |
| 3D model (Jmol) | Interactive image |
| Specific rotation | +110.2° |
| Melting point | 155 °C (311 °F) |
| SEC combustion | −11080 kJ/mol |
| |
| |
| (verify) | |
Testosterone is a medication and naturally occurring steroid hormone.[3] It is used to treat male hypogonadism and certain types of breast cancer. It may also be used to increase athletic ability in the form of doping.[3] It is unclear if the use of testosterone for low levels due to aging is beneficial or harmful.[4] Testosterone can be used as a gel or patch that is applied to the skin, injection into a muscle, tablet that is placed in the cheek, or tablet that is taken by mouth.[3]
Common side effects from testosterone medication include acne, swelling, and breast enlargement in males. Serious side effects may include liver toxicity, heart disease, and behavioral changes. Women and children who are exposed may develop virilization. It is recommended that individuals with prostate cancer not use the medication. It can cause harm if used during pregnancy or breastfeeding.[3]
Testosterone was first isolated in 1935.[5] Rates of use have increased three times in the United States between 2001 and 2011.[6] It is on the WHO Model List of Essential Medicines, the most important medications needed in a basic health system.[7] It is available as a generic medication.[3] The price depends on the form of testosterone used.[8]
Medical uses

The primary use of testosterone is the treatment of males with too little or no natural testosterone production, also termed hypogonadism or hypoandrogenism (androgen deficiency).[9] This treatment is referred to as hormone replacement therapy (HRT), or alternatively, and more specifically, as testosterone replacement therapy (TRT) or androgen replacement therapy (ART). It is used to maintain serum testosterone levels in the normal male range. Decline of testosterone production with age has led to interest in testosterone supplementation.[10]
Deficiency
Testosterone deficiency (also termed hypotestosteronism or hypotestosteronemia) is an abnormally low testosterone production. It may occur because of testicular dysfunction (primary hypogonadism) or hypothalamic-pituitary dysfunction (secondary hypogonadism) and may be congenital or acquired.[11]
Low levels due to aging
Testosterone levels may decline gradually with age.[12][13] The United States Food and Drug Administration (FDA) stated in 2015 that neither the benefits nor the safety of testosterone supplement have been established for low testosterone levels due to aging.[4] The FDA has required that labels on testosterone include warnings about increased risk of heart attacks and stroke.[4]
Transgender men
To take advantage of its virilizing effects, testosterone is administered to transgender men as part of their hormone therapy,[14] with a "target level" of the average male's testosterone level.
Women
Low sexual desire
Testosterone supplementation is effective in the short term for hypoactive sexual desire disorder.[15] However, its long term safety is unclear.[15]
Low testosterone levels
Treating low androgen levels with testosterone is not generally recommended in women when it is due to hypopituitarism, adrenal insufficiency, or following surgical removal of the ovaries.[15] It is also not usually recommended for improving cognition, the risk of heart disease, bone strength or for generalized well being.[15]
Non-medical use
Athletics
Testosterone is used as a form of doping among athletes in order to improve performance.[16] Testosterone is classified as an anabolic agent and is on the World Anti-Doping Agency (WADA) List of Prohibited Substances and Methods.[16] Several application methods for testosterone, including intramuscular injections, transdermal gels and patches, and implantable pellets. Hormone supplements cause the endocrine system to adjust its production and lower the natural production of the hormone, so when supplements are discontinued, natural hormone production is lower than it was originally.
Anabolic-androgenic steroids (AAS), including testosterone and its esters, have also been taken to enhance muscle development, strength, or endurance. They do so directly by increasing the muscles' protein synthesis. As a result, muscle fibers become larger and repair faster than the average person's.
After a series of scandals and publicity in the 1980s (such as Ben Johnson's improved performance at the 1988 Summer Olympics), prohibitions of AAS use were renewed or strengthened by many sports organizations. Testosterone and other AAS were designated a "controlled substance" by the United States Congress in 1990, with the Anabolic Steroid Control Act.[17] Their use is seen as an issue in modern sport, particularly given the lengths to which athletes and professional laboratories go to in trying to conceal such use from sports regulators. Steroid use once again came into the spotlight recently as a result of Canadian professional wrestler Chris Benoit's double murder-suicide in 2007; however, there is no evidence implicating steroid use as a factor in the incident.
Some female athletes may have naturally higher levels of testosterone than others, and may be asked to consent to sex verification and either surgery or drugs to decrease testosterone levels.[18] This has proven contentious, with the Court of Arbitration for Sport suspending the IAAF policy due to insufficient evidence of a link between high androgen levels and improved athletic performance.[19][20]
Detection of abuse
A number of methods for detecting testosterone use by athletes have been employed, most based on a urine test. These include the testosterone/epitestosterone ratio (normally less than 6), the testosterone/luteinizing hormone ratio and the carbon-13/carbon-12 ratio (pharmaceutical testosterone contains less carbon-13 than endogenous testosterone). In some testing programs, an individual's own historical results may serve as a reference interval for interpretation of a suspicious finding. Another approach being investigated is the detection of the administered form of testosterone, usually an ester, in hair.[21][22][23][24]
Contraindications
Absolute contraindications of testosterone include prostate cancer, elevated hematocrit (>54%), uncontrolled congestive heart failure, various other cardiovascular diseases, and uncontrolled obstructive sleep apnea.[25] Breast cancer is said by some sources to be an absolute contraindication of testosterone therapy,[25] but androgens including testosterone have also actually been used to treat breast cancer.[26] Relative contraindications of testosterone include elevated prostate-specific antigen (PSA) in men with a high risk of prostate cancer due to ethnicity or family history, severe lower urinary tract symptoms, and elevated hematocrit (>50%).[25]
Interactions
5α-Reductase inhibitors
5α-Reductase inhibitors like finasteride and dutasteride slightly increase circulating levels of testosterone by inhibiting its metabolism. However, these drugs do this via prevention of the conversion of testosterone into its more potent metabolite dihydrotestosterone (DHT), and this results in strongly reduced circulating levels of DHT (which circulates at much lower relative concentrations). In addition, local levels of DHT in so-called androgenic (5α-reductase-expressing) tissues are also markedly reduced, and this can have a marked impact on certain effects of testosterone. For instance, growth of body and facial hair and penis enlargement induced by testosterone may be inhibited by 5α-reductase inhibitors, and this may be considered undesirable in the context of, for instance, puberty induction. On the other hand, 5α-reductase inhibitors may prevent or reduce adverse androgenic side effects of testosterone like scalp hair loss, oily skin, acne, and seborrhea. In addition to the prevention of testosterone conversion into DHT, 5α-reductase inhibitors also prevent the formation of neurosteroids like 3α-androstanediol from testosterone.
Aromatase inhibitors
Aromatase inhibitors like anastrozole prevent the conversion of testosterone into estradiol by aromatase. As only a very small fraction of testosterone is converted into estradiol, this does not affect testosterone levels, but it can prevent estrogenic side effects that can occur when testosterone is administered at relatively high dosages.
Cytochrome P450 inhibitors
Inhibitors of cytochrome P450 enzymes like the CYP3A4 inhibitor ketoconazole have been associated with only very small effects on circulating testosterone levels.
Antiandrogens and estrogens
Antiandrogens like cyproterone acetate, spironolactone, and bicalutamide can block the androgenic and anabolic effects of testosterone. Estrogens can reduce the effects of testosterone by increasing the hepatic production and in turn circulating levels of sex hormone-binding globulin (SHBG), a carrier protein that binds to and occupies androgens like testosterone and DHT.
Adverse effects
Adverse effects may also include minor side effects such as oily skin, acne, and seborrhea, as well as loss of scalp hair, which may be prevented or reduced with 5α-reductase inhibitors.[27] In women, testosterone can produce hirsutism (excessive facial/body hair growth), deepening of the voice, and other signs of virilization. Exogenous testosterone may cause suppression of spermatogenesis in men, leading to, in some cases, reversible infertility.[28] Gynecomastia and breast tenderness may occur with high dosages of testosterone due to peripheral conversion of testosterone by aromatase into excessive amounts of the estrogen estradiol.[29] Testosterone treatment, particularly in high dosages, can also be associated with mood changes, increased aggression, increased sex drive, spontaneous erections, and nocturnal emissions.[30][31][32][33]
Other side effects include increased hematocrit, which can require venipuncture in order to treat, and exacerbation of sleep apnea.[34]
The FDA stated in 2015 that neither the benefits nor the safety of testosterone have been established for low testosterone levels due to aging.[4] The FDA has required that testosterone pharmaceutical labels include warning information about the possibility of an increased risk of heart attacks and stroke.[4] They have also required the label include concerns about abuse and dependence.[35]
Long-term adverse effects
Cardiovascular disease
Adverse effects of testosterone supplementation may include increased cardiovascular events (including strokes and heart attacks) and deaths based on three peer-reviewed studies involving men taking testosterone replacement.[36] In addition, an increase of 30% in deaths and heart attacks in older men has been reported.[37] Due to an increased incidence of adverse cardiovascular events compared to a placebo group, a Testosterone in Older Men with Mobility Limitations (TOM) trial (a National Institute of Aging randomized trial) was halted early by the Data Safety and Monitoring Committee.[38] On January 31, 2014, reports of strokes, heart attacks, and deaths in men taking FDA-approved testosterone-replacement led the FDA to announce that it would be investigating the issue.[39] Later, in September 2014, the FDA announced, as a result of the "potential for adverse cardiovascular outcomes", a review of the appropriateness and safety of Testosterone Replacement Therapy (TRT).[40][41][42] The FDA now requires warnings in the drug labeling of all approved testosterone products regarding deep vein thrombosis and pulmonary embolism.[43]
Up to the year 2010, studies had not shown any effect on the risk of death, prostate cancer or cardiovascular disease;[44][45] more recent studies, however, do raise concerns.[46] A 2013 study, published in the Journal of the American Medical Association, reported "the use of testosterone therapy was significantly associated with increased risk of adverse outcomes." The study began after a previous, randomized, clinical trial of testosterone therapy in men was stopped prematurely "due to adverse cardiovascular events raising concerns about testosterone therapy safety."[37]
Prostate cancer
Testosterone in the presence of a slow-growing prostate cancer is assumed to increase its growth rate. However, the association between testosterone supplementation and the development of prostate cancer is unproven.[47] Nevertheless, physicians are cautioned about the cancer risk associated with testosterone supplementation.[48]
It may accelerate pre-existing prostate cancer growth in individuals who have undergone androgen deprivation.[34] It is recommended that physicians screen for prostate cancer with a digital rectal exam and prostate-specific antigen (PSA) level before starting therapy, and monitor PSA and hematocrit levels closely during therapy.[12]
Ethnic groups have different rates of prostate cancer.[49] Differences in sex hormones, including testosterone, have been suggested as an explanation for these differences.[49] This apparent paradox can be resolved by noting that prostate cancer is very common. In autopsies, 80% of 80-year-old men have prostate cancer.[50]
Pregnancy and breastfeeding
Testosterone is contraindicated in pregnancy and not recommended during breastfeeding.[51] Androgens like testosterone are teratogens and are known to cause fetal harm, such as producing virilization and ambiguous genitalia.
Pharmacology

Testosterone is a high affinity ligand for and agonist of the nuclear androgen receptor (AR). In addition, testosterone binds to and activates membrane androgen receptors (mARs) such as GPRC6A. Testosterone is also potentiated via transformation by 5α-reductase into the more potent androgen DHT in so-called androgenic tissues such as the prostate gland, seminal vesicles, skin, and hair follicles. In contrast to the case of testosterone, such potentiation occurs to a reduced extent or not at all with most synthetic AAS (as well as with DHT), and this is primarily responsible for the dissociation of anabolic and androgenic effects with these agents.[52] In addition to DHT, testosterone is converted at a rate of approximately 0.3% into the estrogen estradiol via aromatase.[53] This occurs in many tissues, especially adipose tissue, the liver, and the brain, but primarily in adipose tissue.[53] Testosterone, after conversion into DHT, is also metabolized into 3α-androstanediol, a neurosteroid and potent positive allosteric modulator of the GABAA receptor, and 3β-androstanediol, a potent and preferential agonist of the ERβ.[54] Research suggests that these metabolites, along with estradiol, are involved in a number of the effects of testosterone in the brain, including its antidepressant, anxiolytic, stress-relieving, rewarding, and pro-sexual effects.[54]
Pharmacokinetics
Routes of administration
Testosterone is not active orally except in extremely high dosages due to poor absorption and extensive first-pass metabolism.[55] In addition, steroidal androgens including testosterone are hepatotoxic and there is a potential for liver injury with high dosages of oral testosterone due to the production of supraphysiological local concentrations of drug in the liver.[56] Instead of oral ingestion, testosterone is administered parenterally in the form of topical gels and creams, transdermal patches, buccal tablets, and subdermal implants.[55][57] In addition, it is administered via depot intramuscular injection in the form of long-acting ester prodrugs such as testosterone cypionate, testosterone enanthate, and testosterone propionate, as well as the particularly long-lasting testosterone undecanoate.[55] Testosterone buciclate is an even longer-acting testosterone ester that has been developed, but has yet to be approved for medical use.[55]
Although testosterone itself is not used orally, testosterone undecanoate is approved and used orally.[55] Due to the unique chemical properties afforded by its very long ester chain, testosterone undecanoate partially bypasses first-pass liver metabolism via absorption from the gastrointestinal tract directly into the lymphatic system and then into circulation.[55] Of oral testosterone undecanoate that reaches circulation, 90 to 100% is transported lymphatically.[58] This ester is not hepatotoxic at the dosages used.[55] However, oral testosterone undecanoate has variable pharmacokinetics and must be taken two to four times a day with food.[55] In addition to testosterone undecanoate, the combination of testosterone with a 5α-reductase inhibitor like dutasteride can render testosterone orally active when it is given in the form of oil-filled capsules.[59] This is via reduction of the first-pass hepatic metabolism of testosterone.[59]
Absorption
The oral bioavailability of testosterone is very low and virtually negligible.[60][61] The bioavailability of oral testosterone undecanoate is 3 to 7%.[62][58] Topical testosterone gels have a bioavailability of 10% when administered to recommended skins sites including the abdomen, arms, shoulders, and thighs.[63] The bioavailability of testosterone via implant is virtually 100%,[64] while the bioavailability of drugs that are administered intramuscularly is generally almost 95%.[65]
Distribution
In circulation, 97.0 to 99.5% of testosterone is bound to plasma proteins, with 0.5 to 3.0% unbound.[2] It is tightly bound to SHBG and weakly to albumin.[2] Of circulating testosterone, 30 to 44% is bound to SHBG while 54 to 68% is bound to albumin.[2] Testosterone that is unbound is referred to as free testosterone and testosterone that is bound to albumin is referred to as bioavailable testosterone.[2] Unlike testosterone that is bound to SHBG, bioavailable testosterone is bound to plasma proteins weakly enough such that, similarly to free testosterone, it may be biologically active, at least to some extent.[2] When referenced collectively (i.e., free, bioavailable, and SHBG-bound), circulating testosterone is referred to as total testosterone.[2]
Metabolism
Testosterone is metabolized primarily in the liver mainly (90%) by reduction via 5α- and 5β-reductase and conjugation via glucuronidation and sulfation.[2][66][67] The main urinary metabolites of testosterone are androsterone glucuronide and etiocholanolone glucuronide.[2][66][67][68]
Terminal half-life and duration
| Preparation | Route | MRT | t½ |
|---|---|---|---|
| Testosterone undecanoate | p.o. | 3.7 hours | 1.6 hours |
| Testosterone propionate | i.m. | 1.5 days | 0.8 days |
| Testosterone enanthate (in castor oil) | i.m. | 8.5 days | 4.5 days |
| Testosterone undecanoate (in tea seed oil) | i.m. | 34.9 days | 20.9 days |
| Testosterone undecanoate (in castor oil) | i.m. | 36.0 days | 33.9 days |
| Testosterone buciclate (aqueous suspension) | i.m. | 60.0 days | 29.5 days |
The terminal half-life of testosterone varies depending on the route of administration and formulation and on whether or not it is esterified.[60] Oral testosterone undecanoate (in oil capsules) has a terminal half-life of 1.6 hours.[69] Because of its very short terminal half-life, oral testosterone undecanoate is taken two to four times per day.[69] In contrast to oral testosterone, other forms of testosterone including topical gels and solutions, transdermal patches, and buccal tablets have an extended-release effect and can be administered less frequently, at intervals, depending on the route/formulation, of once a day, twice a day, or once every other day.[60][69]
Whereas the terminal half-life of unesterified testosterone administered via intramuscular injection is very short at only around 10 minutes, the terminal half-lives of intramuscular testosterone esters are far longer.[60][69] Administered in the form of oil solutions, the terminal half-lives are 0.8 days for testosterone propionate, 4.5 days for testosterone enanthate, 20.9 days (in tea seed oil) and 33.9 days (in caster oil) for testosterone undecanoate, and 29.5 days for testosterone buciclate.[60][69] Although exact values are not available for intramuscular testosterone cypionate, its pharmacokinetics are said to be the same as those of testosterone enanthate, with "extremely comparable" patterns of testosterone release.[69][71] Due to their varying and different terminal half-lives, the different intramuscular testosterone esters are administered with differing frequencies.[72] Testosterone propionate is injected two to three times per week, testosterone enanthate and testosterone cypionate are injected once every two to four weeks, and testosterone undecanoate and testosterone buciclate are injected once every 10 to 14 weeks.[72] Due to its relatively short duration, testosterone propionate is now relatively little used and testosterone undecanoate is the preferred testosterone ester for intramuscular use.[60][69] Testosterone undecanoate can be injected intramuscularly as little as four times per year.[60][69]
The absorption half-life of subdermal testosterone implants is 2.5 months.[60] The replacement interval is once every four to five months.[73]
Elimination
Testosterone and its metabolites are eliminated in the urine.[74] It is excreted mainly as androsterone glucuronide and etiocholanolone glucuronide.[68] It is also excreted to a small extent as other conjugates such as testosterone glucuronide and androstanediol glucuronides.[68] Only a very small amount of testosterone is found unchanged in the urine.[74]
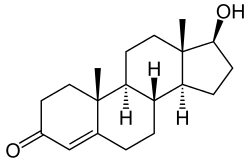
Chemistry
Testosterone is an androstane (C19) steroid and is also known by the chemical name androst-4-en-17β-ol-3-one.[75] It has a double bond between the C4 and C5 positions (making it an androstene), a ketone group at the C3 position, and a hydroxyl (alcohol) group at the C17β position.[75]
Derivatives
Testosterone esters are substituted at the C17β position with a lipophilic fatty acid ester moiety of varying chain length.[76] Major testosterone esters include testosterone cypionate, testosterone enanthate, testosterone propionate, and testosterone undecanoate.[55][75][77] A C17β ether prodrug of testosterone, cloxotestosterone acetate, has also been marketed, although it is little known and is used very rarely or no longer.[75] Another C17β ether prodrug of testosterone, silandrone, also exists but was never marketed, and is notable in that it is orally active.[75] In addition to ester and ether prodrugs, androgen prohormones or precursors of testosterone, such as dehydroepiandrosterone (DHEA), androstenediol, and androstenedione, exist as well, and convert into testosterone to variable extents upon oral ingestion.[78] Unlike testosterone ester and ether prodrugs however, these prohormones are only weakly androgenic/anabolic.[78]
All synthetic AAS are derivatives of testosterone.[78] Prominent examples include nandrolone (19-nortestosterone), metandienone (17α-methyl-δ1-testosterone), and stanozolol (a 17α-alkylated derivative of DHT). Unlike testosterone, AAS that are 17α-alkylated, like metandienone and stanozolol, are orally active. This is due to steric hindrance of C17β-position metabolism during the first-pass through the liver. In contrast, most AAS that are not 17α-alkylated, like nandrolone, are not active orally, and must instead be administered via intramuscular injection. This is almost always in ester form; for instance, in the case of nandrolone, as nandrolone decanoate or nandrolone phenylpropionate.
History
Testosterone was first isolated and synthesized in 1935.[79] Shortly thereafter, in 1937, testosterone first became commercially available as a pharmaceutical drug in the form of pellets and then in ester form for intramuscular injection as the relatively short-acting testosterone propionate.[80][60][71] Methyltestosterone, the first AAS and orally active androgen, was introduced in 1935, but was associated with hepatotoxicity and eventually became largely medically obsolete.[80] In the mid-1950s, the longer-acting testosterone esters testosterone enanthate and testosterone cypionate were introduced.[80] They largely superseded testosterone propionate and became the major testosterone esters used medically for over half a century.[80] In the 1970s, testosterone undecanoate was introduced for oral use in Europe,[80] although intramuscular testosterone undecanoate had already been in use in China for several years.[81] Intramuscular testosterone undecanoate was not introduced in Europe and the United States until much later (in the early to mid 2000s and 2014, respectively).[82][2]
Society and culture
Generic name
Testosterone (pronounced tess-toss-ter-own)[1] is the generic name of testosterone in English and Italian and the INN, USAN, USP, BAN, and DCIT of the drug, while testostérone is its French name and the DCF.[83][84][75] It is also referred to in Latin as testosteronum, in Spanish and Portuguese as testosterona, and in German, Dutch, and Russian and other Slavic languages as testosteron.[83][84] The Cyrillic script of testosterone is тестостерон.[85]
Brand names
Testosterone is marketed under a large number of brand names throughout the world.[83] Major brand names of testosterone and/or its esters include Andriol, Androderm, Androgel, Axiron, Delatestryl, Depo-Testosterone, Intrinsa, Nebido, Omnadren, Primoteston, Sustanon, Testim, Testogel, Testopatch, Testoviron, and Tostran.[83][55][52]
Availability
United States
As of November 2016, unmodified (non-esterified) testosterone is available in the United States in the following formulations:[86]
- Topical gels: Androgel, Fortesta, Testim, Testosterone (generic)
- Topical solutions: Axiron, Testosterone (generic)
- Transdermal patches: Androderm, Testoderm (discontinued), Testoderm TTS (discontinued), Testosterone (generic)
- Intranasal gels: Natesto
- Buccal tablets: Striant
- Pellet implants: Testopel
And the following ester prodrugs of testosterone are available in the United States in oil solutions for intramuscular injection:[86]
- Testosterone cypionate: Depo-Testosterone, Testosterone Cypionate (generic)
- Testosterone enanthate: Delatestryl, Testosterone Enanthate (generic)
- Testosterone propionate: Testosterone Propionate (generic)
- Testosterone undecanoate: Aveed
Unmodified testosterone was also formerly available for intramuscular injection but was discontinued.[86]
Testosterone cypionate and testosterone enanthate were formerly available in combination with estradiol cypionate and estradiol valerate, respectively, under the brand names Depo-Testadiol and Ditate-DS, respectively, as oil solutions for intramuscular injection, but these formulations have been discontinued.[86]
Unlike in Europe, Canada, and much of the rest of the world, oral testosterone undecanoate is not available in the United States.[86][87][33]
Canada
As of November 2016, testosterone is available in Canada in the form of topical gels (Androgel, Testim), topical solutions (Axiron), transdermal patches (Androderm), and intranasal gels (Natesto).[88] Testosterone cypionate (Depo-Testosterone, Testosterone Cypionate (generic)), testosterone enanthate (Delatestryl, PMS-Testosterone Enanthate), and testosterone propionate (Testosterone Propionate (generic)) are available as oil solutions for intramuscular injection and testosterone undecanoate (Andriol, PMS-Testosterone, Taro-Testosterone) is available in the form of oral capsules.[88] Testosterone buccal tablets and pellet implants do not appear to be available in Canada.[88]
Other countries
Testosterone and/or its esters are widely available in countries throughout the world in a variety of formulations.[83]
Legal status
Testosterone and its esters, along with other AAS, are prescription-only controlled substances in many countries throughout the world. In the United States, they are Schedule III drugs under the Controlled Substances Act, in Canada, they are Schedule IV drugs under the Controlled Drugs and Substances Act, and in the United Kingdom, they are Class C drugs under the Misuse of Drugs Act.
Litigation
As of 2014, a number of lawsuits are underway against manufacturers of testosterone, alleging a significantly increased rate of stroke and heart attack in elderly men who use testosterone supplementation.[89]
Research
Depression
Testosterone has been used to treat depression in men who are of middle age with low testosterone. However, a 2014 review showed no benefit on the mood of the men with normal levels of testosterone or on the mood of the older men with low testosterone.[90]
Heart failure
Testosterone replacement can significantly improve exercise capacity, muscle strength and reduce QT intervals in men with chronic heart failure (CHF). Over the 3 to 6 month course of the studies reviewed, testosterone therapy appeared safe and generally effective, and (ruling out prostate cancer) the authors found no justification to absolutely restrict its use in men with CHF.[91] A similar 2012 review also found increased exercise capacity and reasoned the benefits generlizable to women.[92] However, both reviews advocate larger, longer term, randomized controlled trials.[91][92]
Male contraception
Testosterone has been studied and promoted as a male contraceptive analogous to estrogen based contraceptives in women. Otherwise considered an adverse effect of testosterone, reduced spermatogenesis can be further suppressed with the addition of a progestin, improving the contraceptive effect.[93][94]
Miscellaneous
Testosterone therapy may improve the management of type 2 diabetes.[95] Low testosterone has been associated with the development of Alzheimer's disease.[96][97]
References
- 1 2 Susan M. Ford; Sally S. Roach (7 October 2013). Roach's Introductory Clinical Pharmacology. Lippincott Williams & Wilkins. pp. 502–. ISBN 978-1-4698-3214-2.
- 1 2 3 4 5 6 7 8 9 10 Shlomo Melmed; Kenneth S. Polonsky; P. Reed Larsen; Henry M. Kronenberg (11 November 2015). Williams Textbook of Endocrinology. Elsevier Health Sciences. pp. 709,711,765. ISBN 978-0-323-34157-8.
- 1 2 3 4 5 "Testosterone". Drugs.com. American Society of Health-System Pharmacists. December 4, 2015. Retrieved 3 September 2016.
- 1 2 3 4 5 Staff (3 March 2015). "Testosterone Products: Drug Safety Communication – FDA Cautions About Using Testosterone Products for Low Testosterone Due to Aging; Requires Labeling Change to Inform of Possible Increased Risk of Heart Attack And Stroke". FDA. Retrieved 5 March 2015.
- ↑ Taylor WN (2002). Anabolic Steroids and the Athlete (2 ed.). McFarland. p. 180. ISBN 978-0-7864-1128-3.
- ↑ Desroches B, Kohn TP, Welliver C, Pastuszak AW (April 2016). "Testosterone therapy in the new era of Food and Drug Administration oversight". Translational Andrology and Urology. 5 (2): 207–12. doi:10.21037/tau.2016.03.13. PMC 4837303
 . PMID 27141448.
. PMID 27141448. - ↑ "19th WHO Model List of Essential Medicines (April 2015)" (PDF). WHO. April 2015. Retrieved May 10, 2015.
- ↑ Hamilton R (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 197. ISBN 978-1-284-05756-0.
- ↑ Winn, Katherine L. Margo|Robert. "Testosterone Treatments: Why, When, and How? - American Family Physician". www.aafp.org. Retrieved 2016-10-03.
- ↑ Myers JB, Meacham RB (2003). "Androgen replacement therapy in the aging male". Reviews in Urology. 5 (4): 216–26. PMC 1508369
 . PMID 16985841.
. PMID 16985841. - ↑ Gould DC, Petty R (Aug 2000). "The male menopause: does it exist?: for: some men need investigation and testosterone treatment". The Western Journal of Medicine. 173 (2): 76–8. doi:10.1136/ewjm.173.2.76. PMC 1070997
 . PMID 10924412.
. PMID 10924412. - 1 2 Liverman CT, Blazer DG, Institute of Medicine (US) Committee on Assessing the Need for Clinical Trials of Testosterone Replacement Therapy (January 1, 2004). "Introduction". Testosterone and Aging: Clinical Research Directions. National Academies Press (US). doi:10.17226/10852. ISBN 978-0-309-09063-6 – via www.ncbi.nlm.nih.gov.
- ↑ Yeap BB, Almeida OP, Hyde Z, Norman PE, Chubb SA, Jamrozik K, Flicker L (May 2007). "In men older than 70 years, total testosterone remains stable while free testosterone declines with age. The Health in Men Study". European Journal of Endocrinology / European Federation of Endocrine Societies. 156 (5): 585–94. doi:10.1530/EJE-06-0714. PMID 17468195 – via PubMed.
- ↑ "Gender dysphoria – Treatment". NHS Gov.uk. May 21, 2012. Retrieved October 31, 2013.
- 1 2 3 4 Wierman ME, Arlt W, Basson R, Davis SR, Miller KK, Murad MH, Rosner W, Santoro N (Oct 2014). "Androgen therapy in women: a reappraisal: an Endocrine Society clinical practice guideline". The Journal of Clinical Endocrinology and Metabolism. 99 (10): 3489–510. doi:10.1210/jc.2014-2260. PMID 25279570.
- 1 2 "S1. Anabolic Agents | List of Prohibited Substances and Methods". English. Retrieved 2016-06-06.
- ↑ "Anabolic Steroid Control Act" (PDF). United States Sentencing Commission. 1990.
- ↑ Karkazis K, Jordan-Young R (April 11, 2014). "The Trouble With Too Much T". New York Times. Retrieved April 12, 2014.
- ↑ Fagan K (August 13, 2016). "Katie Ledecky is crushing records, so why are we still worried about Caster Semenya?". ESPN. Retrieved 2016-08-27.
- ↑ Padawer, Ruth (June 28, 2016). "The Humiliating Practice of Sex-Testing Female Athletes". The New York Times. ISSN 0362-4331. Retrieved 2016-08-27.
- ↑ Strahm E, Emery C, Saugy M, Dvorak J, Saudan C (Dec 2009). "Detection of testosterone administration based on the carbon isotope ratio profiling of endogenous steroids: international reference populations of professional soccer players". British Journal of Sports Medicine. 43 (13): 1041–44. doi:10.1136/bjsm.2009.058669. PMC 2784500
 . PMID 19549614.
. PMID 19549614. - ↑ Kicman AT, Cowan DA (Jan 2009). "Subject-based profiling for the detection of testosterone administration in sport". Drug Testing and Analysis. 1 (1): 22–4. doi:10.1002/dta.14. PMID 20355155.
- ↑ Pozo OJ, Deventer K, Van Eenoo P, Rubens R, Delbeke FT (Aug 2009). "Quantification of testosterone undecanoate in human hair by liquid chromatography-tandem mass spectrometry". Biomedical Chromatography. 23 (8): 873–80. doi:10.1002/bmc.1199. PMID 19353724.
- ↑ Baselt RC (2008). Disposition of Toxic Drugs & Chemicals in Man (8th ed.). Foster City, Calif: Biomedical Publications. pp. 1501–04. ISBN 978-0-9626523-7-0.
- 1 2 3 Parviz Kavoussi; Raymond A. Costabile; Andrea Salonia (19 October 2012). Clinical Urologic Endocrinology: Principles for Men’s Health. Springer Science & Business Media. pp. 65–. ISBN 978-1-4471-4405-2.
- ↑ Michael Clinton Perry (2008). The Chemotherapy Source Book. Lippincott Williams & Wilkins. pp. 368–. ISBN 978-0-7817-7328-7.
- ↑ Grech A, Breck J, Heidelbaugh J (October 2014). "Adverse effects of testosterone replacement therapy: an update on the evidence and controversy". Therapeutic Advances in Drug Safety. 5 (5): 190–200. doi:10.1177/2042098614548680. PMC 4212439
 . PMID 25360240 – via PubMed Central.
. PMID 25360240 – via PubMed Central. - ↑ "Contraceptive efficacy of testosterone-induced azoospermia in normal men. World Health Organization Task Force on methods for the regulation of male fertility". Lancet. 336 (8721): 955–9. October 1990. doi:10.1016/0140-6736(90)92416-F. PMID 1977002.
- ↑ Rhoden EL, Morgentaler A (February 2004). "Treatment of testosterone-induced gynecomastia with the aromatase inhibitor, anastrozole". International Journal of Impotence Research. 16 (1): 95–7. doi:10.1038/sj.ijir.3901154. PMID 14963480.
- ↑ Yates, William R. (2000). "Testosterone in Psychiatry". Archives of General Psychiatry. 57 (2): 155. doi:10.1001/archpsyc.57.2.155. ISSN 0003-990X.
- ↑ Johnson JM, Nachtigall LB, Stern TA (2013). "The effect of testosterone levels on mood in men: a review". Psychosomatics. 54 (6): 509–14. doi:10.1016/j.psym.2013.06.018. PMID 24016385.
- ↑ Davidson JM, Kwan M, Greenleaf WJ (1982). "Hormonal replacement and sexuality in men". Clin Endocrinol Metab. 11 (3): 599–623. PMID 6814798.
- 1 2 Carrie Bagatell; William J. Bremner (27 May 2003). Androgens in Health and Disease. Springer Science & Business Media. pp. 144,259–261,351. ISBN 978-1-59259-388-0.
- 1 2 Pastuszak AW, Pearlman AM, Lai WS, Godoy G, Sathyamoorthy K, Liu JS, Miles BJ, Lipshultz LI, Khera M (Aug 2013). "Testosterone replacement therapy in patients with prostate cancer after radical prostatectomy". The Journal of Urology. 190 (2): 639–44. doi:10.1016/j.juro.2013.02.002. PMC 4544840
 . PMID 23395803.
. PMID 23395803. - ↑ "Testosterone and Other Anabolic Androgenic Steroids (AAS): FDA Statement - Risks Associated With Abuse and Dependence". FDA. 25 October 2016. Retrieved 26 October 2016.
- ↑ Finkle WD, Greenland S, Ridgeway GK, Adams JL, Frasco MA, Cook MB, Fraumeni JF, Hoover RN (January 2014). "Increased risk of non-fatal myocardial infarction following testosterone therapy prescription in men" (PDF). PLoS ONE. 9 (1): e85805. Bibcode:2014PLoSO...985805F. doi:10.1371/journal.pone.0085805. PMC 3905977
 . PMID 24489673.
. PMID 24489673. - 1 2 Vigen R, O'Donnell CI, Barón AE, Grunwald GK, Maddox TM, Bradley SM, Barqawi A, Woning G, Wierman ME, Plomondon ME, Rumsfeld JS, Ho PM (Nov 2013). "Association of testosterone therapy with mortality, myocardial infarction, and stroke in men with low testosterone levels". JAMA. 310 (17): 1829–36. doi:10.1001/jama.2013.280386. PMID 24193080.
- ↑ Basaria S, Coviello AD, Travison TG, Storer TW, Farwell WR, Jette AM, Eder R, Tennstedt S, Ulloor J, Zhang A, Choong K, Lakshman KM, Mazer NA, Miciek R, Krasnoff J, Elmi A, Knapp PE, Brooks B, Appleman E, Aggarwal S, Bhasin G, Hede-Brierley L, Bhatia A, Collins L, LeBrasseur N, Fiore LD, Bhasin S (2010-07-08). "Adverse events associated with testosterone administration". The New England Journal of Medicine. 363 (2): 109–22. doi:10.1056/NEJMoa1000485. PMC 3440621
 . PMID 20592293.
. PMID 20592293. - ↑ Staff (January 31, 2014). "FDA evaluating risk of stroke, heart attack and death with FDA-approved testosterone products" (PDF). U.S. Food and Drug Administration. Retrieved September 17, 2014.
- ↑ Tavernise S (September 17, 2014). "F.D.A. Panel Backs Limits on Testosterone Drugs". New York Times. Retrieved September 18, 2014.
- ↑ Staff (September 5, 2014). "FDA Panel To Review Testosterone Therapy Appropriateness and Safety". CNN News. Retrieved September 14, 2014.
- ↑ Staff (September 17, 2014). "Joint Meeting for Bone, Reproductive and Urologic Drugs Advisory Committee (BRUDAC) and the Drug Safety And Risk Management Advisory Committee (DSARM AC) – FDA background documents for the discussion of two major issues in testosterone replacement therapy (TRT): 1. The appropriate indicated population for TRT, and 2. The potential for adverse cardiovascular outcomes associated with use of TRT" (PDF). Food and Drug Administration. Retrieved September 14, 2014.
- ↑ Staff (June 19, 2014). "FDA adding general warning to testosterone products about potential for venous blood clots". FDA. Retrieved October 9, 2014.
- ↑ Haddad RM, Kennedy CC, Caples SM, Tracz MJ, Boloña ER, Sideras K, Uraga MV, Erwin PJ, Montori VM (Jan 2007). "Testosterone and cardiovascular risk in men: a systematic review and meta-analysis of randomized placebo-controlled trials". Mayo Clinic Proceedings. 82 (1): 29–39. doi:10.4065/82.1.29. PMID 17285783.
- ↑ Fernández-Balsells MM, Murad MH, Lane M, Lampropulos JF, Albuquerque F, Mullan RJ, Agrwal N, Elamin MB, Gallegos-Orozco JF, Wang AT, Erwin PJ, Bhasin S, Montori VM (Jun 2010). "Clinical review 1: Adverse effects of testosterone therapy in adult men: a systematic review and meta-analysis". The Journal of Clinical Endocrinology and Metabolism. 95 (6): 2560–75. doi:10.1210/jc.2009-2575. PMID 20525906.
- ↑ "Testosterone Products: Drug Safety Communication – FDA Investigating Risk of Cardiovascular Events". FDA. January 31, 2014. Retrieved February 3, 2014.
- ↑ Rhoden EL, Averbeck MA (Nov 2009). "Testosterone therapy and prostate carcinoma". Current Urology Reports. 10 (6): 453–59. doi:10.1007/s11934-009-0072-1. PMID 19863857.
- ↑ Gaylis FD, Lin DW, Ignatoff JM, Amling CL, Tutrone RF, Cosgrove DJ (Aug 2005). "Prostate cancer in men using testosterone supplementation". The Journal of Urology. 174 (2): 534–38; discussion 538. doi:10.1097/01.ju.0000165166.36280.60. PMID 16006887.
- 1 2 Calistro Alvarado L (2010). "Population differences in the testosterone levels of young men are associated with prostate cancer disparities in older men". American Journal of Human Biology. 22 (4): 449–55. doi:10.1002/ajhb.21016. PMID 20087895.
- ↑ Bostwick DG, Burke HB, Djakiew D, Euling S, Ho SM, Landolph J, Morrison H, Sonawane B, Shifflett T, Waters DJ, Timms B (Nov 2004). "Human prostate cancer risk factors". Cancer. 101 (10 Suppl): 2371–490. doi:10.1002/cncr.20408. PMID 15495199. Lay summary – Mercer University School of Medicine.
- ↑ "Testosterone Pregnancy and Breastfeeding Warnings". Retrieved February 1, 2014.
- 1 2 Kicman AT (June 2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–21. doi:10.1038/bjp.2008.165. PMID 18500378.
- 1 2 Burtis CA, Ashwood ER, Bruns DE (14 October 2012). Tietz Textbook of Clinical Chemistry and Molecular Diagnostics. Elsevier Health Sciences. pp. 1947–. ISBN 1-4557-5942-2.
- 1 2 Kohtz, Amy S.; Frye, Cheryl A. (2012). "Dissociating Behavioral, Autonomic, and Neuroendocrine Effects of Androgen Steroids in Animal Models". 829: 397–431. doi:10.1007/978-1-61779-458-2_26. ISSN 1064-3745.
- 1 2 3 4 5 6 7 8 9 10 Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 1116,1119,1182–1185. ISBN 978-0-7817-1750-2.
- ↑ Smith EW, Maibach HI (2 November 2005). Percutaneous Penetration Enhancers, Second Edition. CRC Press. pp. 413–. ISBN 978-1-4200-3920-7.
- ↑ Bandeira F, Gharib H, Golbert A, Griz L, Faria M (26 October 2013). Endocrinology and Diabetes: A Problem-Oriented Approach. Springer Science & Business Media. pp. 88–. ISBN 978-1-4614-8684-8.
- 1 2 Lemke TL, Williams DA (24 January 2012). Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. pp. 1360–. ISBN 978-1-60913-345-0.
- 1 2 Corona G, Rastrelli G, Vignozzi L, Maggi M (2012). "Emerging medication for the treatment of male hypogonadism". Expert Opin Emerg Drugs. 17 (2): 239–59. doi:10.1517/14728214.2012.683411. PMID 22612692.
- 1 2 3 4 5 6 7 8 9 Eberhard Nieschlag; Hermann M. Behre (6 December 2012). Testosterone: Action - Deficiency - Substitution. Springer Science & Business Media. pp. 9,309–331,350. ISBN 978-3-642-72185-4.
- ↑ Steven B. Karch, MD, FFFLM (21 December 2006). Drug Abuse Handbook, Second Edition. CRC Press. pp. 700–. ISBN 978-1-4200-0346-8.
- ↑ Touitou E, Barry BW (27 November 2006). Enhancement in Drug Delivery. CRC Press. pp. 122–. ISBN 978-1-4200-0481-6.
- ↑ Jones H (25 September 2008). Testosterone Deficiency in Men. OUP Oxford. pp. 89–. ISBN 978-0-19-954513-1.
- ↑ Bhasin S (13 February 1996). Pharmacology, Biology, and Clinical Applications of Androgens: Current Status and Future Prospects. John Wiley & Sons. pp. 462–. ISBN 978-0-471-13320-9.
- ↑ Conceptual Pharmacology. Universities Press. 2010. pp. 8–. ISBN 978-81-7371-679-9.
- 1 2 Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 1116, 1119, 1183. ISBN 978-0-7817-1750-2.
- 1 2 Wecker L, Watts S, Faingold C, Dunaway G, Crespo L (1 April 2009). Brody's Human Pharmacology. Elsevier Health Sciences. pp. 468–469. ISBN 0-323-07575-4.
- 1 2 3 Thieme D, Hemmersbach P (18 December 2009). Doping in Sports. Springer Science & Business Media. pp. 53–. ISBN 978-3-540-79088-4.
- 1 2 3 4 5 6 7 8 9 Eberhard Nieschlag; Hermann M. Behre; Susan Nieschlag (13 January 2010). Andrology: Male Reproductive Health and Dysfunction. Springer Science & Business Media. pp. 441–446. ISBN 978-3-540-78355-8.
- ↑ Behre HM, Abshagen K, Oettel M, Hübler D, Nieschlag E (1999). "Intramuscular injection of testosterone undecanoate for the treatment of male hypogonadism: phase I studies". Eur. J. Endocrinol. 140 (5): 414–9. PMID 10229906.
- 1 2 William Llewellyn (2011). Anabolics. Molecular Nutrition Llc. pp. 413,426,607. ISBN 978-0-9828280-1-4.
- 1 2 Sai-Ching Jim Yeung; Carmen P. Escalante; Robert F. Gagel (2009). Medical Care of Cancer Patients. PMPH-USA. pp. 247–. ISBN 978-1-60795-008-0.
- ↑ Parveen Kumar; Michael L Clark (4 June 2012). Kumar and Clark's Clinical Medicine. Elsevier Health Sciences. pp. 976–. ISBN 0-7020-5304-X.
- 1 2 Karch SB, Drummer O (26 December 2001). Karch's Pathology of Drug Abuse (third ed.). CRC Press. pp. 486–. ISBN 978-1-4200-4211-5.
- 1 2 3 4 5 6 J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 641–642. ISBN 978-1-4757-2085-3.
- ↑ J. Larry Jameson; Leslie J. De Groot (25 February 2015). Endocrinology: Adult and Pediatric. Elsevier Health Sciences. pp. 2387–. ISBN 978-0-323-32195-2.
- ↑ Christopher R. Chapple; William D. Steers (10 May 2011). Practical Urology: Essential Principles and Practice: Essential Principles and Practice. Springer Science & Business Media. pp. 228–. ISBN 978-1-84882-034-0.
- 1 2 3 Haff , G. Gregory; Triplett , N. Travis (23 September 2015). Essentials of Strength Training and Conditioning 4th Edition. Human Kinetics. pp. 229,233. ISBN 978-1-4925-0162-6.
- ↑ Taylor WN (16 January 2002). Anabolic Steroids and the Athlete (2nd ed.). McFarland. pp. 180–. ISBN 978-0-7864-1128-3.
- 1 2 3 4 5 Hoberman J (21 February 2005). Testosterone Dreams: Rejuvenation, Aphrodisia, Doping. University of California Press. pp. 134–. ISBN 978-0-520-93978-3.
- ↑ Anthony R. Mundy; John Fitzpatrick; David E. Neal; Nicholas J. R. George (26 July 2010). The Scientific Basis of Urology. CRC Press. pp. 294–. ISBN 978-1-84184-749-8.
- ↑ Profile, A. R. D. (2004). Testosterone Undecanoate—Schering AG. Drugs, 5(6), 368-369. http://link.springer.com/article/10.2165/00126839-200405060-00012
- 1 2 3 4 5 "Testosterone - International - Drugs.com". Drugs.com. Retrieved 12 November 2016.
- 1 2 Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. ISBN 978-3-88763-075-1.
- ↑ Владимир Мюллер (15 April 2016). Англо-русский словарь. Русско-английский словарь. 250 000 слов. ЛитРес. pp. 643–. ISBN 978-5-457-98308-3.
- 1 2 3 4 5 "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 16 November 2016.
- ↑ John E. Morley; Lucretia van den Berg (5 November 1999). Endocrinology of Aging. Springer Science & Business Media. pp. 141–. ISBN 978-1-59259-715-4.
- 1 2 3 "Drug Product Database - Health Canada". Health Canada. Retrieved 13 November 2016.
- ↑ Harris A. "Abbott Labs Sued by Five Men Claiming Androgel Injuries". Bloomberg.com. Bloomberg, L.P. Retrieved June 16, 2014.
- ↑ Amanatkar HR, Chibnall JT, Seo BW, Manepalli JN, Grossberg GT (Feb 2014). "Impact of exogenous testosterone on mood: a systematic review and meta-analysis of randomized placebo-controlled trials". Annals of Clinical Psychiatry. 26 (1): 19–32. PMID 24501728.
- 1 2 Wang W, Jiang T, Li C, Chen J, Cao K, Qi LW, Li P, Zhu W, Zhu B, Chen Y (May 2016). "Will testosterone replacement therapy become a new treatment of chronic heart failure? A review based on 8 clinical trials". Journal of Thoracic Disease. 8 (5): E269–77. doi:10.21037/jtd.2016.03.39. PMC 4842839
 . PMID 27162680.
. PMID 27162680. - 1 2 Toma M, McAlister FA, Coglianese EE, Vidi V, Vasaiwala S, Bakal JA, Armstrong PW, Ezekowitz JA (May 2012). "Testosterone Supplementation in Heart Failure: A Meta-Analysis". Circulation. Heart Failure. 5 (3): 315–21. doi:10.1161/CIRCHEARTFAILURE.111.965632. PMID 22511747.
- ↑ Wang C, Festin MP, Swerdloff RS (2016). "Male Hormonal Contraception: Where Are We Now?". Current Obstetrics and Gynecology Reports. 5: 38–47. doi:10.1007/s13669-016-0140-8. PMC 4762912
 . PMID 26949570.
. PMID 26949570. - ↑ Chao JH, Page ST (July 2016). "The current state of male hormonal contraception". Pharmacology & Therapeutics. 163: 109–17. doi:10.1016/j.pharmthera.2016.03.012. PMID 27016468.
- ↑ Traish AM, Saad F, Guay A (2009). "The dark side of testosterone deficiency: II. Type 2 diabetes and insulin resistance". Journal of Andrology. 30 (1): 23–32. doi:10.2164/jandrol.108.005751. PMID 18772488.
- ↑ Pike CJ, Rosario ER, Nguyen TV (Apr 2006). "Androgens, aging, and Alzheimer's disease". Endocrine. 29 (2): 233–41. doi:10.1385/ENDO:29:2:233. PMID 16785599.
- ↑ Rosario ER, Chang L, Stanczyk FZ, Pike CJ (Sep 2004). "Age-related testosterone depletion and the development of Alzheimer disease". JAMA. 292 (12): 1431–32. doi:10.1001/jama.292.12.1431-b. PMID 15383512.