Norvinisterone
 | |
| Clinical data | |
|---|---|
| Trade names | Nor-Progestelea |
| ATC code | None |
| Identifiers | |
| |
| Synonyms | Neoprogestin, vinylnortestosterone, SC-4641 |
| CAS Number | 6795-60-4 |
| PubChem (CID) | 65588 |
| ChemSpider | 59030 |
| Chemical and physical data | |
| Formula | C20H28O2 |
| Molar mass | 300.435 g/mol |
| 3D model (Jmol) | Interactive image |
| Melting point | 169 to 171 °C (336 to 340 °F) [1] |
| |
| |
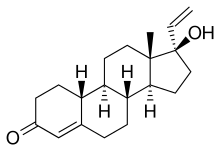
Norvinisterone (INN) (marketed as Neoprogestin and Nor-Progestelea by Syntex; former developmental code name SC-4641), also known as 17α-vinyl-19-nortestosterone (or vinylnortestosterone), is a steroidal progestin and androgenic-anabolic steroid (AAS) of the 19-nortestosterone group that is or has been used in Europe.[1][2][3][4][5] It was synthesized in 1953,[2] and was studied in humans by 1960.[6] It appears to be quite androgenic, with about one-third and one-fifth of the androgenic and anabolic activity, respectively, of nandrolone (19-nortestosterone) in animal bioassays.[7]
See also
References
- 1 2 Merck Index, 11th edition, 6637
- 1 2 J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 889–. ISBN 978-1-4757-2085-3.
- ↑ Pei-Show Juo (21 December 2001). Concise Dictionary of Biomedicine and Molecular Biology. CRC Press. pp. 774–. ISBN 978-1-4200-4130-9.
- ↑ P. H. List; L. Hörhammer (12 March 2013). Chemikalien und Drogen Teil A: N-Q. Springer Berlin Heidelberg. pp. 274–. ISBN 978-3-642-65035-2.
- ↑ Meyerson, Bengt J. (1967). "Relationship Between the Anesthetic and Gestagenic Action and Estrous Behavior-Inducing Activity of Different Progestins". Endocrinology. 81 (2): 369–374. doi:10.1210/endo-81-2-369. ISSN 0013-7227.
- ↑ MARTINEZ MONTES EA, BAGNATI EP, ZAPATA AC, BUR GE (1960). "[Clinical trial of a new luteoid: norvinisterone]". Dia Med (in Spanish; Castilian). 32: 194–7. PMID 14421807. CS1 maint: Unrecognized language (link)
- ↑ Saunders, Francis J.; Drill, Victor A. (1956). "THE MYOTROPHIC AND ANDROGENIC EFFECTS OF 17-ETHYL-19-NORTESTOSTERONE AND RELATED COMPOUNDS". Endocrinology. 58 (5): 567–572. doi:10.1210/endo-58-5-567. ISSN 0013-7227.
| Androgens (incl. AAS) |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Antiandrogens |
| ||||||||||||||||
See also: Estrogens and antiestrogens • Progestogens and antiprogestogens • Glucocorticoids and antiglucocorticoids • Mineralocorticoids and antimineralocorticoids • Gonadotropins and GnRH | |||||||||||||||||
| PR |
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| mPRs (PAQRs) |
| ||||||||||
See also: Androgenics • Estrogenics • Glucocorticoidics • Mineralocorticoidics • Steroid hormone metabolism modulators | |||||||||||
| AR |
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| GPRC6A |
| ||||||||||
See also: Estrogenics • Glucocorticoidics • Mineralocorticoidics • Progestogenics • Steroid hormone metabolism modulators • List of androgens/anabolic steroids | |||||||||||
