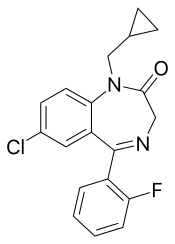
Flutoprazepam
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Restas |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral, Intravenous |
| ATC code | none |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 80-90% |
| Metabolism | Hepatic |
| Biological half-life | 60-90 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number |
25967-29-7 |
| PubChem (CID) | 3400 |
| ChemSpider |
3283 |
| UNII |
2GHY1101MM |
| KEGG |
D01279 |
| ChEMBL | CHEMBL2106743 |
| Chemical and physical data | |
| Formula | C19H16ClFN2O |
| Molar mass | 342.795 |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Flutoprazepam (Restas) is a drug which is a benzodiazepine. It was patented in Japan by Sumitomo in 1972[1] and its medical use remains mostly confined to that country. Its muscle relaxant properties are approximately equivalent to those of diazepam - however, it has more powerful sedative, hypnotic, anxiolytic and anticonvulsant effects and is around four times more potent by weight compared to diazepam.[2] It is longer acting than diazepam due to its long-acting active metabolites,[3] which contribute significantly to its effects.[4]
Flutoprazepam is typically used for the treatment of severe insomnia and may also be used for treating stomach ulcers.[5]
Flutoprazepam is currently a Schedule IV drug under the international Convention on Psychotropic Substances of 1971.[6][7]
- In Singapore, flutoprazepam is a Class C-Schedule II drug under the Misuse of Drugs Act.
- In Thailand, flutoprazepam is a Schedule III psychotropic substance.
- In Hong Kong, flutoprazepam is regulated under Schedule 1 of Hong Kong's Chapter 134 Dangerous Drugs Ordinance. Flutoprazepam can only be used legally by health professionals and for university research purposes. The substance can be given by pharmacists under a prescription. Anyone who supplies the substance without prescription can be fined $10000 (HKD). The penalty for trafficking or manufacturing the substance is a $5,000,000 (HKD) fine and life imprisonment. Possession of the substance for consumption without license from the Department of Health is illegal with a $1,000,000 (HKD) fine and/or 7 years of jail time.[8]
See also
References
- ↑ US patent 3632574, Hisao Yamamoto et al, "PROCESS FOR PRODUCING BENZODIAZEPINE DERIVATIVES", published 1968-29-04, issued 1972-04-01
- ↑ Sukamoto, T.; Aikawa, K.; Itoh, K.; Nose, T. (Sep 1980). "[Psycopharmacological and general pharmacological studies of 7-chloro-1-cyclopropylmethyl-1, 3-dihydro-5-(2-fluorophenyl)-2H-1, 4-benzodiazepin-2-one (KB-509) (author's transl)]". Nippon Yakurigaku Zasshi. 76 (6): 447–68. doi:10.1254/fpj.76.447. PMID 7203280.
- ↑ Ueki, S.; Sukamoto, T.; Watanabe, S.; Yamamoto, T.; Kataoka, Y.; Shibata, S.; Suwandi, D.; Shibata, K.; et al. (Jul 1982). "[Behavioral effects of flutoprazepam (KB-509) and its metabolites]". Nippon Yakurigaku Zasshi. 80 (1): 15–30. doi:10.1254/fpj.80.15. PMID 6890927.
- ↑ Barzaghi, N.; Leone, L.; Monteleone, M.; Tomasini, G.; Perucca, E. (1989). "Pharmacokinetics of flutoprazepam, a novel benzodiazepine drug, in normal subjects.". Eur J Drug Metab Pharmacokinet. 14 (4): 293–8. doi:10.1007/BF03190114. PMID 2633923.
- ↑ Fukuda, T.; Itoh, K.; Nose, T. (Mar 1981). "[Antiulcerogenic action of 7-chloro-1-cyclopropylmethyl-1,3-dihydro-5-(2-fluorophenyl)-2H-1,4-benzodiazepin-2-one (KB-509), a new benzodiazepine derivative]". Nippon Yakurigaku Zasshi. 77 (3): 273–80. doi:10.1254/fpj.77.273. PMID 7052359.
- ↑ http://www.cnb.gov.sg/ [Central Narcotics Bureau, Singapore]
- ↑ "Green List—List of psychotropic substances under international control" (PDF) (23rd ed.). International Narcotics Control Board. August 2003. Retrieved 2007-11-25.
- ↑ "Bilingual Laws Information System" (English). The Government of the Hong Kong Special Administrative Region of the People's Republic of China.