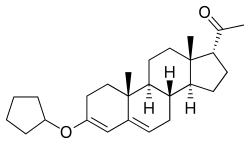
Quingestrone
 | |
| Clinical data | |
|---|---|
| Trade names | Enol-Luteovis |
| Routes of administration | Oral |
| ATC code | G03A (WHO) |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| Synonyms | W-3399; Progesterone 3-cyclopentyl enol ether; 3-Cyclopentyloxypregna-3,5-dien-20-one |
| CAS Number | 67-95-8 |
| PubChem (CID) | 9929903 |
| ChemSpider | 8105534 |
| UNII | 0612VZ9I5Z |
| KEGG | D05680 |
| ChEMBL | CHEMBL2104568 |
| Chemical and physical data | |
| Formula | C26H38O2 |
| Molar mass | 382.57872 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
Quingestrone (INN, USAN) (brand name Enol-Luteovis; former developmental code name W-3399), also known as progesterone 3-cyclopentyl enol ether (PCPE) or as 3-cyclopentyloxypregna-3,5-dien-20-one, is a semisynthetic, steroidal, pure progestin that was developed by Vister and introduced in Italy as an oral contraceptive (in combination with ethinyl estradiol or mestranol)[1][2] in 1962.[3][4][5][6][7] It is closely related to progesterone, being the 3-cyclopentyl enol ether of the progestogen sex hormone.[6]
Pharmacology
Along with the retroprogesterone derivative dydrogesterone, quingestrone is described as a "true" progesterone derivative due to its close similarity to natural progesterone.[8] Similarly to progesterone, dydrogesterone, and hydroxyprogesterone caproate, quingestrone is a pure progestogen and lacks any androgenic effects.[9] As such, it poses no risk of androgenic side effects or virilizing teratogenic effects on female fetuses.[9] Accordingly, the drug was studied in the clinical prevention of miscarriage during pregnancy; however, insufficient efficacy was observed at the dosage assessed (100 mg/day orally).[10][11][12][13] Quingestrone is said to influence the hypothalamic-pituitary-adrenal axis similarly to progesterone and medroxyprogesterone acetate, producing adrenal suppression at sufficiently high doses, and this suggests that it possesses weak or very weak glucocorticoid properties similarly to progesterone.[14][15]
Pharmacokinetics
Quingestrone has been suggested to act as a prodrug of progesterone via slow hydrolysis in the body.[10][16] Indeed, it produces similar metabolites (e.g., pregnanediols and allopregnanediols) to progesterone,[10][17] although with differing ratios,[18][5] and notably is the only progestin that is known to produce pregnanediol as a metabolite.[19] Subsequent research has cast doubt on the notion that quingestrone is a prodrug of progesterone however, and indicates that it instead is directly metabolized into pregnanediols without intermediate conversion into progesterone.[20]
Relative to progesterone, quingestrone shows improved pharmacokinetics, including higher potency,[21] oral activity,[22] and a longer half-life and hence duration of action.[16] This is considered to be due to its higher lipophilicity,[16] being stored into and slowly released from fat.[6][10] Quingestrone also shows slower metabolism and more stable blood levels, with a longer time to peak concentrations and a less intense peak compared to progesterone.[23]
The bioavailability of quingestrone is highest when it is given as a sesame seed oil solution (compared to an oil suspension (~2-fold less) or micronization (~7-fold less)).[17]
See also
- Pentagestrone acetate
- Quingestanol acetate
- Progestogen ester
- 3α-Dihydroprogesterone
- 3β-Dihydroprogesterone
References
- ↑ Larrey D, Geneve J, Pessayre D, Machayekhi JP, Degott C, Benhamou JP (1987). "Prolonged cholestasis after cyproheptadine-induced acute hepatitis". J. Clin. Gastroenterol. 9 (1): 102–4. doi:10.1097/00004836-198702000-00026. ISSN 0192-0790. PMID 3559100.
- ↑ International Planned Parenthood Federation. Medical Committee. Oral Advisory Group (1965). Handbook on oral contraception. Little, Brown. p. 18. OCLC 2717593.
- ↑ J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. p. 163. doi:10.1007/978-1-4757-2085-3. ISBN 978-1-4757-2085-3. OCLC 898564124.
- ↑ Pei-Show Juo (21 December 2001). Concise Dictionary of Biomedicine and Molecular Biology. CRC Press. pp. 922–. ISBN 978-1-4200-4130-9. LCCN 2001043892. OCLC 48057162.
- 1 2 Denis Frank Hawkins (1974). Obstetric therapeutics: clinical pharmacology and therapeutics in obstetric practice. Baillière Tindall. pp. 129, 138, 145. ISBN 978-0-7020-0471-1.
- 1 2 3 P. J. Bentley (1980). Endocrine Pharmacology: Physiological Basis and Therapeutic Applications. CUP Archive. pp. 274–. ISBN 978-0-521-22673-8.
- ↑ Watts, GeorgeT. (1962). "EMERGENCY TREATMENT OF POISONING". The Lancet. 279 (7228): 533–534. doi:10.1016/S0140-6736(62)91504-0. ISSN 0140-6736.
- ↑ Erica G. Wachtel (1969). Exfoliative cytology in gynaecological practice. Appleton-Century-Crofts. p. 134. LCCN 77008744.
- 1 2 Dugald Baird; John Martin Munro Kerr (1969). Combined textbook of obstetrics and gynæcology for students and practitioners. E. & S. Livingstone. LCCN 70360656.
- 1 2 3 4 Burton, Eunice R.; Wachtel, Erica G. (1967). "A CLINICAL TRIAL AND CYTOLOGICAL ASSESSMENT OF ENOL LUTEOVIS IN THE TREATMENT OF THREATENED AND RECURRENT ABORTION". BJOG: An International Journal of Obstetrics and Gynaecology. 74 (4): 533–536. doi:10.1111/j.1471-0528.1967.tb03986.x. ISSN 1470-0328. PMID 5340429.
- ↑ Vitamins and Hormones. Academic Press. 9 February 1973. pp. 332–. ISBN 978-0-08-086627-7.
- ↑ Ian S. Fraser (1998). Estrogens and Progestogens in Clinical Practice. Churchill Livingstone. ISBN 978-0-443-04706-0.
- ↑ Goldstein, Peter; Berrier, Jayne; Rosen, Scott; Sacks, Henry S.; Chalmers, Thomas C. (1989). "A meta-analysis of randomized control trials of progestational agents in pregnancy". BJOG: An International Journal of Obstetrics and Gynaecology. 96 (3): 265–274. doi:10.1111/j.1471-0528.1989.tb02385.x. ISSN 1470-0328. PMID 2653414.
- ↑ Luciano Martini (1966). Neuroendocrinology. Academic Press. p. 331. LCCN 66026256.
- ↑ Steinetz, B.G.; Beach, V.L.; DiPasquale, G.; Battista, J.V. (1965). "Effects of different gestagenic steroid types on plasma-free corticosteroid levels in ACTH-treated rats". Steroids. 5 (1): 93–108. doi:10.1016/0039-128X(65)90134-0. ISSN 0039-128X.
- 1 2 3 Charman, William N.; Porter, Christopher J.H. (1996). "Lipophilic prodrugs designed for intestinal lymphatic transport". Advanced Drug Delivery Reviews. 19 (2): 149–169. doi:10.1016/0169-409X(95)00105-G. ISSN 0169-409X.
- 1 2 Fatouros DG, Karpf DM, Nielsen FS, Mullertz A (2007). "Clinical studies with oral lipid based formulations of poorly soluble compounds". Ther Clin Risk Manag. 3 (4): 591–604. PMC 2374933
 . PMID 18472981.
. PMID 18472981. - ↑ http://www.popline.org/node/474452
- ↑ Current Medicine and Drugs. 1962. ISSN 0590-4048.
Enol Luteovis (3 cyclo-pentyl enol ether of progesterone) is the only oral progestin producing pregnanediol as a metabolite. It is not very potent and probably carries very little risk of producing virilizing effects on a female foetus. Thus it is more closely related to progesterone than the other synthetic progestins.
- ↑ Meli, A.; Wolff, A.; Lucker, W. E.; Steinetz, B. G. (1965). "The Biological Profile of Progesterone 3-Cyclopentyl Enol Ether as Compared with That of Progesterone". Experimental Biology and Medicine. 118 (3): 714–717. doi:10.3181/00379727-118-29947. ISSN 1535-3702.
- ↑ http://www.popline.org/node/474452
- ↑ Joseph Bolivar De Lee (1965). The ... Year Book of Obstetrics and Gynecology. Year Book Publishers. p. 150. ISSN 0084-3911. LCCN cdr38000020.
- ↑ Caie, E.; Klopper, A. (1964). "THE URINARY EXCRETION OF PREGNANEDIOL AFTER THE ADMINISTRATION OF AN ORAL GESTAGEN (PROGESTERONE CYCLOPENTYL ENOL ETHER)". Journal of Endocrinology. 28 (2): 221–222. doi:10.1677/joe.0.0280221. ISSN 0022-0795. PMID 14112260.