Procaterol
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | Oral (tablets, syrup), inhalation (DPI) |
| ATC code | R03AC16 (WHO) R03CC08 (WHO) |
| Identifiers | |
| |
| CAS Number |
59828-07-8 |
| PubChem (CID) | 4916 |
| ChemSpider |
599984 |
| UNII |
X7I3EMM5K0 |
| KEGG |
D08424 |
| ECHA InfoCard | 100.069.606 |
| Chemical and physical data | |
| Formula | C16H22N2O3 |
| Molar mass | 290.357 g/mol |
| 3D model (Jmol) | Interactive image |
| Chirality | Racemic mixture |
| |
| | |
Procaterol, applied as procaterol hydrochloride (USAN), is an intermediate-acting β2 adrenoreceptor agonist used for the treatment of asthma. It has never been filed for FDA evaluation in the United States, where it is not marketed. The drug is readily oxidized in the presence of moisture and air, making it unsuitable for therapeutic use by inhalation.[1] Pharmaceutical company Parke-Davis/Warner-Lambert researched a stabilizer to prevent oxidation, but an effective one was never developed.[1]
Synthesis
Like pirbuterol, procaterol exhibits similar broncholytic properties as salbutamol (albuterol), but it has somewhat of a more prolonged action. It is recommended for use as an inhaled drug for treating asthma.
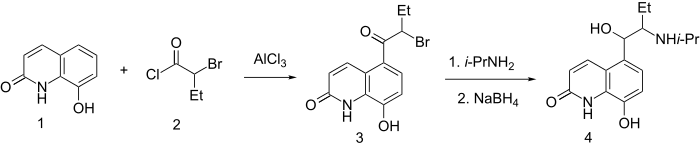
8-Hydroxycarbostyril 1 is acylated with 2-bromobutyric acid chloride 2 at the fifth position of the quinoline system, which gives the compound 3. This undergoes action of isopropylamine, forming an aminoketone, the carbonyl group of which is reduced by sodium borohydride, giving procaterol 4.
Trade names
Procaterol is available under a number of trade names (Onsukil, Masacin, Procadil and others), the most common seems to be Meptin ((KR), (CN), (ID), (MY), (PH), (SG), (TH), (HK)).[6]
References
- 1 2 US, "Procaterol stabilization", published 1984, issued 1986
- ↑ K. Nakagawa, S. Yoshizaki, U.S. Patent 4,026,897 (1977).
- ↑ S. Yoshizaki, M. Osaki, K. Nakagawa, Y. Yamura, Chem. Pharm. Bull., 28, 3441 (1980).
- ↑ Yoshizaki, Shiro; Tanimura, Kaoru; Tamada, Shigeharu; Yabuuchi, Youichi; Nakagawa, Kazuyuki (1976). "Sympathomimetic amines having a carbostyril nucleus". Journal of Medicinal Chemistry. 19 (9): 1138. doi:10.1021/jm00231a011. PMID 10441.
- ↑ Yoshizaki, Shiro; Manabe, Yoshiaki; Tamada, Shigeharu; Nakagawa, Kazuyuki; Tei, Seiso (1977). "Isomers of erythro-5-(1-hydroxy-2-isopropylaminobutyl)-8-hydroxycarbostyril, a new bronchodilator". Journal of Medicinal Chemistry. 20 (8): 1103. doi:10.1021/jm00218a024. PMID 894683.
- ↑ "International Drugs: Procaterol". Drugs.com. Retrieved 7 March 2016.