Curcumin
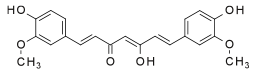 Enol form | |
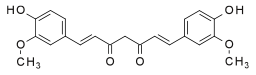 Keto form | |
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(1E,6E)-1,7-Bis(4-hydroxy-3-methoxyphenyl)hepta-1,6-diene-3,5-dione | |
| Other names
(1E,6E)-1,7-Bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione Diferuloylmethane Curcumin I C.I. 75300 Natural Yellow 3 | |
| Identifiers | |
| 458-37-7 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:3962 |
| ChEMBL | ChEMBL116438 |
| ChemSpider | 839564 |
| E number | E100 (colours) |
| 7000 | |
| PubChem | 969516 |
| UNII | IT942ZTH98 |
| |
| |
| Properties | |
| C21H20O6 | |
| Molar mass | 368.39 g·mol−1 |
| Appearance | Bright yellow-orange powder |
| Melting point | 183 °C (361 °F; 456 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Curcumin (/ˈkərkjuːmən/, diferuloylmethane) is a bright yellow chemical produced by some plants. It is the principal curcuminoid of turmeric (Curcuma longa), a member of the ginger family (Zingiberaceae). It is sold as an herbal supplement, cosmetics ingredient, food flavoring and food coloring.[1] As a food additive, its E number is E100.[2]
It was isolated in 1815 when Vogel and Pelletier reported the isolation of a “yellow coloring-matter” from the rhizomes of turmeric and named it curcumin.[3] Although curcumin has been used historically in Ayurvedic medicine,[4] its potential medicinal properties remain unproven and are an area of active investigation.[5]
Chemically, curcumin is a diarylheptanoid, belonging to the group of curcuminoids, which are natural phenols responsible for turmeric's' yellow color. It is a tautomeric compound existing in enolic form in organic solvents and as a keto form in water.[6]
Applications
Food additive/supplement
The most common applications are as a dietary supplement, in cosmetics, as a food coloring and as flavoring for foods such as turmeric-flavored beverages (Japan).[1]

Annual sales of curcumin have increased since 2012, largely due to an increase in its popularity as a dietary supplement.[1] It is increasingly popular in skin care products that are marketed as containing natural ingredients or dyes, especially in Asia.[1] The largest market is in North America, where sales exceeded US$20 million in 2014.[1]
Chemistry
Curcumin incorporates several functional groups whose structure was first identified in 1910.[7] The aromatic ring systems, which are phenols, are connected by two α,β-unsaturated carbonyl groups. The diketones form stable enols and are readily deprotonated to form enolates; the α,β-unsaturated carbonyl group is a good Michael acceptor and undergoes nucleophilic addition.
Curcumin is used as an indicator for boron.[8] It reacts with boric acid to form a red-color compound, rosocyanine.
Biosynthesis
The biosynthetic route of curcumin is uncertain. In 1973, Roughly and Whiting proposed two mechanisms for curcumin biosynthesis. The first mechanism involves a chain extension reaction by cinnamic acid and 5 malonyl-CoA molecules that eventually arylized into a curcuminoid. The second mechanism involves two cinnamate units coupled together by malonyl-CoA. Both use cinnamic acid as their starting point, which is derived from the amino acid phenylalanine.
Plant biosyntheses starting with cinnamic acid is rare compared to the more common p-coumaric acid.[9] Only a few identified compounds, such as anigorufone and pinosylvin, build from cinnamic acid.[10][11]
An experimentally backed route was not presented until 2008. This route follows both Roughley and Whiting mechanisms. However, the labeling data supported the first mechanism model in which 5 malonyl-CoA molecules react with cinnamic acid to form curcumin. However, the sequencing in which the functional groups, the alcohol and the methoxy, introduce themselves onto the curcuminoid seemed to support more strongly the second proposed mechanism.[9] Therefore, the second pathway was accepted.
Pharmacodynamics
In vitro, curcumin has been shown to inhibit certain epigenetic enzymes (the histone deacetylases: HDAC1, HDAC3, and HDAC8) and transcriptional co-activator proteins (the p300 histone acetyltransferase).[12][13][14] Curcumin also inhibits the arachidonate 5-lipoxygenase enzyme in vitro.[15]
Pharmacokinetics
In Phase I clinical trials, dietary curcumin was shown to exhibit poor bioavailability, exhibited by rapid metabolism, low levels in plasma and tissues, and extensive rapid excretion, factors that make its in vivo activity poorly understood.[16] Potential factors that limit the bioavailability of curcumin include insolubility in water (more soluble in alkaline solutions) and non-absorption.[17] Numerous approaches to increase curcumin bioavailability are under research, including the use of absorption factors (such as piperine), liposomes, a structural analogue, or nanomaterials using specialized polymers.[17]
Research
Data from anti-cancer research on curcumin conducted by Bharat Aggarwal, formerly a researcher at the MD Anderson Cancer Center, was deemed fraudulent and subsequently retracted by the publisher.[18]
Two preliminary clinical studies in cancer patients consuming high doses of curcumin (up to 8 grams per day for 3–4 months) showed no toxicity, though some subjects reported mild nausea or diarrhea.[19]
Although curcumin has been assessed in numerous laboratory and clinical studies, it has no medical uses established by well-designed clinical research.[20]
References
- 1 2 3 4 5 Majeed, Shaheen (28 December 2015). "The State of the Curcumin Market". Natural Products Insider.
- ↑ European Commission. "Food Additives". Retrieved 2014-02-15.
- ↑ Vogel, H.; Pelletier, J. (1815). "Curcumin-biological and medicinal properties". Journal de Pharmacie. I: 289.
- ↑ Wilken, Reason; Veena, Mysore S.; Wang, Marilene B.; Srivatsan, Eri S. (2011). "Curcumin: A review of anti-cancer properties and therapeutic activity in head and neck squamous cell carcinoma". Molecular Cancer. 10: 12. doi:10.1186/1476-4598-10-12. ISSN 1476-4598. PMC 3055228
 . PMID 21299897.
. PMID 21299897. - ↑ "Turmeric". US National Center for Complementary and Integrative Health, National Institutes of Health. 31 May 2016. Retrieved 15 June 2016.
- ↑ Manolova, Y.; Antonov, L.; Deneva, V. at al (2014). "The effect of the water on the curcumin tautomerism: A quantitative approach". Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. 132: 815–820. doi:10.1016/j.saa.2014.05.096.
- ↑ Miłobȩdzka, J.; v. Kostanecki, St.; Lampe, V. (1910). "Zur Kenntnis des Curcumins". Berichte der deutschen chemischen Gesellschaft. 43 (2): 2163–70. doi:10.1002/cber.191004302168.
- ↑ "EPA Method 212.3: Boron (Colorimetric, Curcumin)" (PDF).
- 1 2 3 Kita, Tomoko; Imai, Shinsuke; Sawada, Hiroshi; et al. (2008). "The Biosynthetic Pathway of Curcuminoid in Turmeric (Curcuma longa) as Revealed by 13C-Labeled Precursors". Bioscience, Biotechnology, and Biochemistry. 72 (7): 1789–1798. doi:10.1271/bbb.80075.
- ↑ Schmitt, Bettina; Hölscher, Dirk; Schneider, Bernd (2000). "Variability of phenylpropanoid precursors in the biosynthesis of phenylphenalenones in Anigozanthos preissii". Phytochemistry. 53 (3): 331–7. doi:10.1016/S0031-9422(99)00544-0. PMID 10703053.
- ↑ Gehlert, R.; Schoeppner, A.; Kindl, H. (1990). "Stilbene Synthase from Seedlings of Pinus sylvestris: Purification and Induction in Response to Fungal Infection" (pdf). Molecular Plant-Microbe Interactions. 3 (6): 444–449. doi:10.1094/MPMI-3-444.
- ↑ Reuter, S; Gupta, SC; Park, B; et al. (May 2011). "Epigenetic changes induced by curcumin and other natural compounds". Genes Nutr. 6 (2): 93–108. doi:10.1007/s12263-011-0222-1. PMC 3092901
 . PMID 21516481.
. PMID 21516481.
Figure 2 - ↑ Vahid, F; Zand, H; Nosrat-Mirshekarlou, E; et al. (May 2015). "The role dietary of bioactive compounds on the regulation of histone acetylases and deacetylases: a review". Gene. 562 (1): 8–15. doi:10.1016/j.gene.2015.02.045. PMID 25701602.
- ↑ "Curcumin". IUPHAR. IUPHAR/BPS Guide to PHARMACOLOGY. Retrieved 22 May 2015.
- ↑ Bishayee, K; Khuda-Bukhsh, AR (September 2013). "5-lipoxygenase antagonist therapy: a new approach towards targeted cancer chemotherapy". Acta Biochim. Biophys. Sin. (Shanghai). 45 (9): 709–719. doi:10.1093/abbs/gmt064. PMID 23752617.
- ↑ Devassy, JG; Nwachukwu, ID; Jones, PJ (March 2015). "Curcumin and cancer: barriers to obtaining a health claim". Nutrition Reviews. 73 (3): 155–65. doi:10.1093/nutrit/nuu064. PMID 26024538.
- 1 2 Yallapu MM, Jaggi M, Chauhan SC (2012). "Curcumin nanoformulations: a future nanomedicine for cancer". Drug Discov Today. 17 (1-2): 71–80. doi:10.1016/j.drudis.2011.09.009. PMC 3259195
 . PMID 21959306.
. PMID 21959306. - ↑ Ackerman T (29 February 2012). "M.D. Anderson professor under fraud probe". Houston Chronicle. Retrieved 8 March 2016.
- ↑ Hsu, C. H.; Cheng, A. L. (2007). "Clinical studies with curcumin". Advances in Experimental Medicine and Biology. 595: 471–480. doi:10.1007/978-0-387-46401-5_21. ISBN 978-0-387-46400-8. PMID 17569225.
- ↑ "Curcumin". Micronutrient Information Center; Phytochemicals. Linus Pauling Institute, Oregon State University, Corvallis. 2016. Retrieved 18 June 2016.
External links
- Turmeric and curcumin, from Memorial Sloan-Kettering Cancer Center
- Turmeric, from the University of Maryland Medical Center
