Netupitant
 | |
| Clinical data | |
|---|---|
| Trade names | Akynzeo |
| Routes of administration | Oral |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | >60% (estimated) |
| Protein binding | >99% |
| Metabolism | mainly CYP3A4; also CYP2D6 and CYP2C9 |
| Biological half-life | 88 hours |
| Excretion | 71% (faeces) |
| Identifiers | |
| |
| CAS Number | 290297-26-6 |
| PubChem (CID) | 6451149 |
| DrugBank | DB09048 |
| ChemSpider | 4953629 |
| UNII | 7732P08TIR |
| KEGG | D05152 |
| ChEBI | CHEBI:85155 |
| ChEMBL | CHEMBL206253 |
| Chemical and physical data | |
| Formula | C30H32F6N4O |
| Molar mass | 578.59 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
Netupitant is an antiemitic drug. In the United States, the combination drug netupitant/palonosetron (trade name Akynzeo) is approved by the Food and Drug Administration for prevention of acute and delayed chemotherapy-induced nausea and vomiting, including highly emetogenic chemotherapy such as with cisplatin.[1] In Europe, it is approved by the European Medicines Agency for the same indication.[2]
Adverse effects
Side effects of the combination netupitant/palonosetron are similar to palonosetron alone, so that no common side effects can be attributed to netupitant.[2]
Interactions
Netupitant blood plasma levels are expected to increase when combined with inhibitors of the liver enzyme CYP3A4 and lowered when combined with inductors of this enzyme.[2]
Being a CYP3A4 inhibitor itself, netupitant could also increase plasma levels of pharmaceuticals that are metabolized by CYP3A4. This effect has been observed with dexamethasone, the anti-cancer drugs docetaxel and etoposide, and to a minor (not clinically significant) extent with levonorgestrel, erythromycin and midazolam.[2]
Pharmacology
Mechanism of action
Netupitant is a selective NK1 receptor antagonist.[3]
Pharmacokinetics
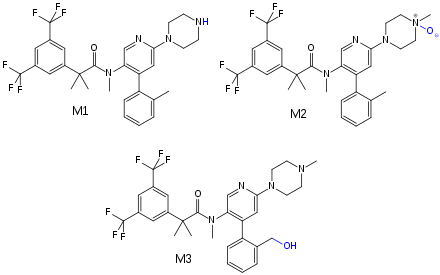
Bioavailability is estimated to be over 60% for orally taken netupitant. Highest blood plasma concentrations are reached five hours after application. Availability is moderately (10–20%) increased when taken after a fatty meal. Netupitant and its main metabolites (called M1 and M3) are bound to plasma proteins to more than 99%, and M2 protein binding is 97%.[2]
The substance is mainly metabolized by CYP3A4, and to a lesser extent by CYP2D6 and CYP2C9. The main metabolites are desmethyl-netupitant (M1), netupitant N-oxide (M2), and hydroxy-netupitant (M3); all three are pharmacologically active.[2][4]
Netupitant and its metabolites are mainly excreted via the faeces.[2] Biological half-life is 88 hours, significantly longer than that of the first NK1 receptor antagonist, aprepitant, which has a half-life of 9 to 13 hours.[5]
References
- ↑ "FDA approves Akynzeo for nausea and vomiting associated with cancer chemotherapy". Food and Drug Administration. October 10, 2014.
- 1 2 3 4 5 6 7 "Akynzeo: Summary of Product Characteristics" (PDF). European Medicines Agency. Retrieved 12 July 2016.
- ↑ Rizzi A, Campi B, Camarda V, Molinari S, Cantoreggi S, Regoli D, Pietra C, Calo G (2012). "In vitro and in vivo pharmacological characterization of the novel NK(1) receptor selective antagonist netupitant". Peptides. 37 (1): 86–97. doi:10.1016/j.peptides.2012.06.010. PMID 22732666.
- 1 2 Spinelli, T; Calcagnile, S; Giuliano, C; Rossi, G; Lanzarotti, C; Mair, S; Stevens, L; Nisbet, I (2013). "Netupitant PET imaging and ADME studies in humans". The Journal of Clinical Pharmacology. 54 (1): 97–108. doi:10.1002/jcph.198. PMC 4282341
 .
. - ↑ Haberfeld, H, ed. (2015). Austria-Codex (in German). Vienna: Österreichischer Apothekerverlag.