Oenanthotoxin
 | |
| Names | |
|---|---|
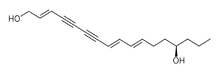
| IUPAC name
(2E,8E,10E,14R)-Heptadeca-2,8,10-triene-4,6-diyne-1,14-diol | |
| Other names
Enanthotoxin | |
| Identifiers | |
| 20311-78-8 | |
| 3D model (Jmol) | Interactive image |
| ChEMBL | ChEMBL550225 |
| ChemSpider | 24616929 |
| KEGG | C20044 |
| PubChem | 6436464 |
| |
| |
| Properties | |
| C17H22O2 | |
| Molar mass | 258.36 g·mol−1 |
| Melting point | 86 °C (187 °F; 359 K) |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
| LD50 (median dose) |
0.58 mg/kg for mice |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Oenanthotoxin is a toxin extracted from hemlock water dropwort (Oenanthe crocata) and other plants of the genus Oenanthe. It is a central nervous system poison, and acts as a noncompetitive gamma-aminobutyric acid (GABA) antagonist.[1] A case has been made for the presence of this toxin in local Oenanthe species playing a causative role in euthanasia in ancient Sardinia.[2][3] It was crystallized in 1949 by Clarke and co-workers.[4] It is structurally closely related to the toxins cicutoxin[5] and carotatoxin.[6][7] Oenanthotoxin is a C17 polyacetylene isomer of cicutoxin.
Occurrence
Oenanthotoxin concentration in plants is dependent on seasonal changes and geographical location, the most is present during late winter and early spring.[8] Contrary to most poisonous plants that contain bitter tastes or burning sensations, the water dropwort has a rather sweet and pleasant taste and odor.[9] Water dropwort is characterized by a yellow liquid that changes color due to air exposure.[1][9] The roots are the most toxic part, although the entire plant contains poisonous properties.[8] Unlike the majority of poisonous plants, the poisonous properties are not lost due to plant drying.[10]
History and culture
The discovery and use of plants containing oenanthotoxin predates Socrates and Homer and its first use as a poison is thought to have been implemented between 1800 BCE and 800 BCE in Pre-Roman Sardinia.[9][11] In Ancient Sardinia, it was considered to be a humane form of euthanasia. Elderly people who were unable to care for themselves were given water dropwort and dropped from a high rock to ensure death.[9][11] It is also believed that Socrates ingested the plant to commit suicide.[12]
A common symptom of oenanthotoxin is risus sardonicus, better known as the Sardonic Grin, coined by Homer in the 8th century BCE, due to the victim's rigid smile after ingestion.
Furthermore, as a muscle relaxant, it is believed to have cosmetic botox-like properties in small amounts.[11]
Mechanism of action
Although oenanthotoxin is a relatively well known poison, its mechanism of action is not entirely understood. However, there is evidence that it is similar to the mechanism of cicutoxin.
Oenanthotoxin is part of a group of C17 conjugated polyacetylenes that act as noncompetitive gamma-aminobutyric acid (GABA) inhibitors in the central nervous system (CNS). GABA binds to the beta-domain of the GABAA receptor in the CNS and activates the receptor causing chloride ions to flow across the membrane which hyperpolarizes the neuron.[1] When oenanthotoxin is introduced to the body, it non-competitively binds to the same beta-domain receptor as GABA would have and prevents normal inhibitory function. Binding to the same receptor, oenanthotoxin blocks the chloride channel, allowing depolarization to continue. This causes hyperactivity in the neurons, resulting in convulsions and seizures and thus blocking GABAergic responses.[9]
Symptoms
While oenanthotoxin is extremely dangerous and toxic (LD50 = 0.58 mg/kg for mice),[1] there have been numerous case studies documenting the common symptoms including: convulsions, seizures, nausea, diarrhea, tachycardia, mydriasis, rhabdomyolysis, renal failure, respiratory impairment, and cardiac dysrhythmias.[1][8][9]
Below is a comprehensive table listing the recorded symptoms caused oenanthotoxin within each system in the body Oenanthe crocata:[1]
| Organ system | Symptoms |
|---|---|
| Neurological | slurred speech, dizziness, paresthesia, delirium, ataxia, coma, seizures, trismus, hyperreflexia, opisthotonus, spasms, cerebral edema, status epilepticus |
| Gastrointestinal | nausea, vomiting, salivation, abdominal pain |
| Respiratory | congestion, distress, depression, airway obstruction, arrest, apnea |
| Cardivascular | tachicardia, brachycardia, hypertension, hypotension, cardiac dysrhythmias, cardiac arrest |
| Renal | glycosuria, proteinuria, hematuria, oliguria, myoglobinuria, acute renal failure |
| Musculoskeletal | weakness, muscle spasms, muscle rigidity, rhabdomyolysis |
| Metabolic | elevated temperature, liver dysfunction, hypokalemia, lactic dehydrogenase, disseminating (intravascular, coagulation), metabolic acidosis, azotemia |
| Occular | mydriasis |
| Dermal | diaphoresis, cyanosis, flushed face |
References
- 1 2 3 4 5 6 Schep, L. J.; Slaughter, R. J.; Becket, G.; Beasley, D. M. G. (2009). "Poisoning due to Water Hemlock". Clinical Toxicology. 47 (4): 270–278. doi:10.1080/15563650902904332. PMID 19514873.
- ↑ Appendino, G.; Pollastro, F.; Verotta, L.; Ballero, M.; Romano, A.; Wyrembek, P.; Szczuraszek, K.; Mozrzymas, J. W.; Taglialatela-Scafati, O. (2009). "Polyacetylenes From Sardinian Oenanthe fistulosa: A Molecular Clue to risus sardonicus". Journal of Natural Products. 72 (5): 962–965. doi:10.1021/np8007717. PMC 2685611
 . PMID 19245244.
. PMID 19245244. - ↑ Choi, C. Q.; Harmon, K.; Matson, J. (August 2009). "News Scan Briefs: Killer Smile". Scientific American.
- ↑ E. G. C. Clarke, D. E. Kidder and W. D. Robertson (1949) J. Pharm. Pharmacol. 1 377-381.
- ↑ Anet, E. F. L. J.; Lythgoe, B.; Silk, M. H.; Trippett, S. (1953). "Oenanthotoxin and Cicutoxin. Isolation and Structures". Journal of the Chemical Society. 1953: 309–322. doi:10.1039/JR9530000309.
- ↑ King, L. A.; Lewis, M. J.; Parry, D.; Twitchett, P. J.; Kilner, E. A. (1985). "Identification of Oenanthotoxin and Related Compounds in Hemlock Water Dropwort Poisoning". Human Toxicology. 4 (4): 355–364. doi:10.1177/096032718500400401. PMID 4018815.
- ↑ Anet, E. F. L. J.; Lythgoe, B.; Silk, M. H.; Trippett, S. (1952). "The Chemistry of Oenanthotoxin and Cicutoxin". Chemistry and Industry. 31: 757–758.
- 1 2 3 "Information Sheet: 31 Hemlock Water Dropwort (Oenanthe crocata)" (PDF). Centre for Ecology & Hydrology. Centre for Aquatic Plant Management. Retrieved 2005. Check date values in:
|access-date=(help) - 1 2 3 4 5 6 Appendino, G.; Pollastro, F.; Verotta, L. (2009), "Polyacetylenes from Sardinian Oenanthe Fistulosa: A Molecular Clue to risus sardonicus", J. Nat. Prod., 72: 962–965, doi:10.1021/np8007717, PMC 2685611
 , PMID 19245244
, PMID 19245244 - ↑ Egdahl, A. (1911). "A case of poisoning due to eating poison hemlock (Cicuta maculata) with a review of reported cases". Arch Intern Med. 7: 348–356. doi:10.1001/archinte.1911.00060030061002.
- 1 2 3 Owen, James. "Ancient Death-Smile Potion Decoded?". National Geographic. Journal of Natural Products. Retrieved June 2, 2009.
- ↑ Bletchly, Rachael. "Killers in your garden; Beware these poison plants". The Free Library. Gale, Cengage Learning.
