Tolcapone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Tasmar |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a698036 |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | N04BX01 (WHO) |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 65-85% |
| Protein binding | >99.9% |
| Metabolism | Hepatic (glucuronidation) |
| Biological half-life | 2-3 hours |
| Excretion | Urine (60%), faeces (40%) |
| Identifiers | |
| |
| CAS Number |
134308-13-7 |
| PubChem (CID) | 4659569 |
| IUPHAR/BPS | 6646 |
| DrugBank |
DB00323 |
| ChemSpider |
3848682 |
| UNII |
CIF6334OLY |
| KEGG |
D00786 |
| ChEMBL |
CHEMBL1324 |
| ECHA InfoCard | 100.222.604 |
| Chemical and physical data | |
| Formula | C14H11NO5 |
| Molar mass | 273.241 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Tolcapone (brand name Tasmar) is a drug used to treat Parkinson's disease (PD). It is a selective, potent and reversible nitro catechol type inhibitor of enzyme catechol-O-methyl transferase (COMT).[1]
Uses
Tolcapone is used in the treatment of Parkinson's disease as an adjunct to levodopa/carbidopa medication that is conventionally paired with an aromatic L-amino acid decarboxylase (AADC) inhibitor.[3]
Normally, administration of levodopa is compromised when COMT converts it to 3-methoxy-DOPA. By preventing this effect, more of the levodopa that is administered reaches the CNS. Additionally, levodopa that is in the CNS, after being converted to dopamine, will not be degraded by COMT when tolcapone inhibits COMT activity.
Chemistry
Tolcapone is a yellow, odorless, non-hygroscopic, crystalline compound with a relative molecular mass of 273.25 g/mol. The chemical name of tolcapone is 3,4-dihydroxy-4’-methyl-5-nitrobenzophenone. Its empirical formula is C14H11NO5.
History
Tolcapone was introduced into European Market August 1997 and subsequently into the United States Market in March 1998. Liver toxicity or hepatoxicity was reported in four people who were administered tolcapone, 3 people died due to complications. Consequentially, the marketing authorization of tolcapone was suspended from December, 1998 until August, 2004 when it was lifted. In November 1998, the company that manufactured tolcapone voluntarily[4] removed the drug from the market. The authorization was then renewed on August, 2009.[5]
As a result of reported complications, the U.S. Food and Drug Administration (FDA) issued a black-box warning for tolcapone and label revisions that aimed to regulate the monitoring of those prescribed tolcapone for PD in November 1998.[6] A number of other countries withdrew tolcapone from the market; Australia in February 1999, Bulgaria in April 1999, Iceland in November 1998, Lithuania in December 1998.[4]
Pharmacodynamics
Tolcapone binds to the catalytic center of COMT in both peripheral and central nervous systems with greater affinity than any of the three catecholamine, including levodopa.[7] It thereby prevents the 3-0-methylation of L-DOPA (3-hydroxy-L-tyrosine) by COMT which produces 3-O-methyldopa, a major metabolite that competes with levodopa to cross the blood-brain barrier. Thus, tolcapone improves the bioavailability and reduces the clearance of levodopa and subsequently dopamine from the central nervous system (CNS).[8]

The strength of the binding affinity of tolcapone, represented by the inhibition constant Ki (2.50 nM), can be thought of as the dissociation constant for enzyme and inhibitor complex kinetics. Maximum catalytic activity denotes the efficacy of tolcapone (Vmax=58.4 pmol/min·mg).[9]
Pharmacokinetics
Typical dose of tolcapone is 100 or 200 mg, orally administered three times daily.[10] The half life of tolcapone is 2–3 hours, volume of distribution (Vd) being 0.3 L/kg (21 L in average 70 kg person) and bioavailability about 60%.[11]
Tolcapone has the ability to cross the blood–brain barrier and thus exerts its COMT inhibitory effects in the central nervous system (CNS) as well as in the periphery.
99% of tolcapone is in monoanionic form when in body because the physiological pH is 7.4. Tolcapone penetrates the blood-brain barrier better than two other nitrocatechols such as nitecapone and entacapone because it has higher lipophilicity due to its R substituent. Partition coefficients quantify the ability of the molecule to cross the blood-brain barrier. LogPIdce= 0.2, -1.4, -0.4 for tolcapone, nitecapone and entacopone respectively. Partition coefficients in this case were measured in 1,2-dichloroethane / H2O solution which caused molecules to be in ionized form. There is no current explanation for how these charged molecules permeate the blood-brain barrier.[12]
Treatment
Tolcapone is administered adjunct with levodopa and AADC because it increases the bioavailability of levodopa.[8] Since tolcapone has a greater affinity for COMT, it inhibits the enzyme from methylating levodopa. This results in greater levels of levodopa and increases the time period before clearance.
Tolcapone helps alleviates the issues with levodopa 50% of PD patients have experienced. Without administration of tolcapone, the beneficial effects of levodopa have worn off resulting in motor fluctuations.[11]
In comparison with entacapone, another nitro catechol COMT inhibitor, tolcapone has longer half life (2.9 hours vs. 0.8 hours), and can better penetrate the blood-brain barrier in that it acts both centrally and peripherally.[13] However, entacapone is less toxic in regard to liver function.
Adverse effects
Tolcapone has demonstrated significant hepatotoxicity[14] that limits the drug's utility. Entacapone, another COMT inhibitor, is an alternative selection for levodopa adjunct therapy in the treatment of Parkinson's disease, largely since it has a more favorable toxicity profile.
This hepatoxicity can be attributed to elevated levels of transaminases, but studies have shown that minimal risk exists for those without preexisting liver conditions when their enzyme levels were being monitored. No clear mechanism is implicated in tolcapone induced liver toxicity, but it has been hypothesized that it has something to do with abnormal mitochondrial respiration due to the uncoupling of oxidative phosphorylation.[10]
Other side effects that result from the administration of tolcapone regard the increase in dopaminergic activity, digestive symptoms and liver function. Treatment with tolcapone runs the risk of eliciting or prolonging dyskinesia; this can be counteracted by decreasing the dose of levodopa. This occurs because the administration of tolcapone results in the accumulation of the biological methyl donor S-adenosyl-L-methionine (SAM) in the striatum that works to induce symptoms of PD.[10]
Digestive symptoms include nausea, diarrhea, orthostatic hypotension, urine discoloration and dizziness.[10] Tolcapone causes more severe diarrhea than entacapone.
Synthesis
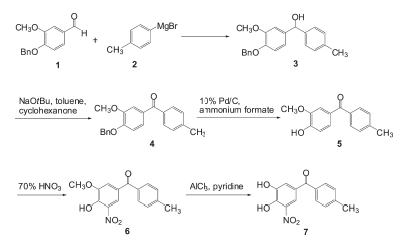
A synthesis of tolcapone proposed in 2008 begins with a Grignard reaction between a benzaldehyde derivative and p-tolyl magnesium bromide. The alcohol thus produced is then converted to a ketone using sodium t-butoxide. The benzyl protecting group is removed by palladium-catalyzed hydrogenation in the presence of ammonium formate. A nitro group is introduced at the 5-position adjacent to the hydroxyl group unmasked in the cleavage of the benzyl ether. The synthesis ends with cleavage of the methoxy group using aluminum chloride to yield the product alcohol.[15]
See also
References
- ↑ Antonini, A.; Abbruzzese, G.; Barone, P.; Bonuccelli, U.; Lopiano, L.; Onofrj, M.; Zappia, M.; Quattrone, A. (2008). "COMT inhibition with tolcapone in the treatment algorithm of patients with Parkinson's disease (PD): Relevance for motor and non-motor features". Neuropsychiatric Disease and Treatment. 4 (1): 1–9. doi:10.2147/NDT.S2404. PMC 2515921
 . PMID 18728767.
. PMID 18728767. - ↑ Ellermann, M.; Lerner, C.; Burgy, G.; Ehler, A.; Bissantz, C.; Jakob-Roetne, R.; Paulini, R.; Allemann, O.; et al. (2012). "Catechol-O-methyltransferase in complex with substituted 3′-deoxyribose bisubstrate inhibitors". Acta Crystallographica D. 68 (3): 253–260. doi:10.1107/S0907444912001138. PMID 22349227.
- ↑ Dingemanse, J.; Jorga, K.; Zurcher, G.; Schmitt, M.; Sedek, G.; Prada, M.; Brummelen, P. (1995). "Pharmacokinetic-pharmacodynamic interaction between the COMT inhibitor tolcapone and single-dose levodopa". British Journal of Clinical Pharmacology. 40 (3): 253–262. doi:10.1111/j.1365-2125.1995.tb05781.x. PMC 1365105
 . PMID 8527287.
. PMID 8527287. - 1 2 "2001 Pharmaceuticals: Restrictions in Use and Availability". World Health Organization. Retrieved 2012-10-31.
- ↑ "Tasmar tolcapone EPAR summary for the public" (PDF). European Medicine Agency. Retrieved 2012-10-31.
- ↑ "FDA Talk Paper". New Warnings for Parkinson's Drug, Tasmar. Food and Drug Administration.
|first1=missing|last1=in Authors list (help) - ↑ Deleu, D.; Northway, M. G.; Hanssens, Y. (2002). "Clinical Pharmacokinetic and Pharmacodynamic Properties of Drugs Used in the Treatment of Parkinsons Disease". Clinical Pharmacokinetics. 41 (4): 261–309. doi:10.2165/00003088-200241040-00003. PMID 11978145.
- 1 2 Jorga, K.; Fotteler, B.; Heizmann, P.; Gasser, R. (2001). "Metabolism and excretion of tolcapone, a novel inhibitor of catechol-O-methyltransferase". British Journal of Clinical Pharmacology. 48 (4): 513–520. doi:10.1046/j.1365-2125.1999.00036.x. PMC 2014389
 . PMID 10583021.
. PMID 10583021. - ↑ Forsberg, M. M.; Huotari, M.; Savolainen, J.; Männistö, P. T. (2005). "The role of physicochemical properties of entacapone and tolcapone on their efficacy during local intrastriatal administration". European Journal of Pharmaceutical Sciences. 24 (5): 503–511. doi:10.1016/j.ejps.2005.01.005. PMID 15784340.
- 1 2 3 4 Truong, D. (2009). "Tolcapone: Review of its pharmacology and use as adjunctive therapy in patients with Parkinson's disease". Clinical Interventions in Aging. 2009 (4): 109–113. doi:10.2147/CIA.S3787. PMC 2685232
 . PMID 19503773.
. PMID 19503773. - 1 2 Jorga, K. M.; Fotteler, B.; Heizmann, P.; Zürcher, G. (1998). "Pharmacokinetics and pharmacodynamics after oral and intravenous administration of tolcapone, a novel adjunct to Parkinson's disease therapy". European Journal of Clinical Pharmacology. 54 (5): 443–447. doi:10.1007/s002280050490. PMID 9754991.
- ↑ Novaroli, L.; Bouchard Doulakas, G.; Reist, M.; Rolando, B.; Fruttero, R.; Gasco, A.; Carrupt, P.-A. (2006). "The Lipophilicity Behavior of Three Catechol-O-methyltransferase (COMT) Inhibitors and Simple Analogues". Helvetica Chimica Acta. 89 (1): 144–152. doi:10.1002/hlca.200690007.
- ↑ Forsberg, M.; Lehtonen, M.; Heikkinen, M.; Savolainen, J.; Järvinen, T.; Männistö, P. T. (2003). "Pharmacokinetics and Pharmacodynamics of Entacapone and Tolcapone after Acute and Repeated Administration: A Comparative Study in the Rat". Journal of Pharmacology and Experimental Therapeutics. 304 (2): 498–506. doi:10.1124/jpet.102.042846. PMID 12538800.
- ↑ Olanow, C. W.; Watkins, P. B. (2007). "Tolcapone". Clinical Neuropharmacology. 30 (5): 287–294. doi:10.1097/wnf.0b013e318038d2b6. PMID 17909307.
- ↑ Manikumar, G.; Jin, C.; Rehder, K. S. (2008). "Convenient Synthesis of Tolcapone, a Selective Catechol‐O‐methyltransferase Inhibitor". Synthetic Communications. 38 (5): 810. doi:10.1080/00397910701821077.
External links
- Tolcapone bound to proteins in the PDB