Piophilidae
| Cheese flies | |
|---|---|
 | |
| Piophila casei | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Diptera |
| Section: | Schizophora |
| Subsection: | Acalyptratae |
| Superfamily: | Tephritoidea |
| Family: | Piophilidae |
| Subfamilies | |
| |
| Synonyms | |
| |
The Piophilidae are a family of "true flies", in the order Diptera. The so-called cheese flies are the best-known members, but most species of the Piophilidae are scavengers in animal products, carrion, and fungi. They may accordingly be important in forensic entomology[1] and medical entomology.[2] For a fly maggot, the larvae of many species have an unusually well-developed ability to leap when alarmed or when abandoning their larval food to pupate; they accordingly may be known as cheese skippers or other kinds of skippers according to their food source.[3]
Overview
The most notorious member of the family is the cheese fly, Piophila casei; it is cosmopolitan, and a typical member of the family. It is a small species, about 4 mm (0.16 in) long. The fly's larvae infest cured meats, smoked or salted fish, cheeses, and carrion. The mature larva is about 8 mm (0.31 in) long and is sometimes called the cheese skipper because of its leaping ability - when disturbed, this tiny maggot can hop some 15 cm (6 in) into the air.[4] Adults are also known as bacon flies and their larvae as bacon skippers, ham skippers, cheese maggots, cheese hoppers, etc. In the Mediterranean island of Sardinia, the larvae are intentionally introduced into pecorino cheese to produce the characteristic casu marzu ("rotten cheese" in Sardinian).
The adult cheese fly's body is black, blue-black, or bronze, with some yellow on the head, antennae, and legs. The wings are faintly iridescent and lie flat upon the fly's abdomen when at rest. At 4 mm (0.16 in) long, the fly is one-third to one-half as long as the common housefly.
Behaviour patterns
Like the larvae of various fly families, including the family Tephritidae, the larvae of typical piophilids are notorious for jumping or "skipping", especially in their final instar.[5] The larvae accomplish their jumps by bending over, grabbing onto the rears of their own bodies with their mouth hooks, and tensing their muscles in a manner that increases the pressure on their own blood and internal organs. When they release their grip, the internal pressure straightens out the tubular body, propelling the forequarters upwards, the rest of the body following.[4] A series of photos illustrating this remarkable behaviour and a video can be seen at . Jumping is performed most typically when the larva is alarmed by a disturbance, or when it is abandoning its feeding site in preparation for pupation.
The tiny piophilid species Protopiophila litigata, commonly known as the antler fly,[6] fly breeds on discarded antlers of moose and other deer. As adaptations to such a scarce and scattered resource, they have several unusual behaviour patterns. For one thing, the males are astonishingly bellicose; the specific epithet litigata reflects the fact — in context it means "aggressive", as in the English word "litigious". On discarded antlers, the males form complex, highly structured aggregations in which a great deal of territorial competition occurs. Some individuals defend stable territories, while others simply wander in search of females that arrive on antlers to feed, mate, and oviposit. In prime areas of the antler, near oviposition sites (cracks in the antler surface), males spend much of their time battling rival males. They even attack insects much larger than themselves.[7] Another adaptation to life on discarded antlers is an astonishing degree of site fidelity: males spend their entire lives competing on the same antler (only leaving to spend the night in nearby vegetation), making it possible to mark flies individually and obtain longitudinal field data on these tiny insects. This unique ecology made it possible to document senescence in wild insects for the first time.[8]
The waltzing fly, Prochyliza xanthostoma, occurs in North America. It is one of the carrion-feeding piophilids and is remarkable for its sexual dimorphism and its patterns of behavioural adaptation and associated morphological adaptations. In particular, the antennae, forelegs, and heads of the males are adapted in unusual ways to their behaviour in combat and courtship.[9]
Waltzing flies breed in early spring, generally on the carcasses of moose. By midspring of a good year, they are one of the most abundant insect species in the local forests. Nubile females bask on vegetation around carcasses, and males aggregate in their sun to court and to defend territories. Competing males usually begin by spreading their forelegs and holding each other's foretarsi. This recalls the competitive behaviour and adaptations of another fly family, the Diopsidae, in which the males match the spread of each other's eyes. Such behaviour may permit the adversaries to assess each other's size, thereby avoiding futile and possibly damaging conflict with a larger opponent. When neither of two competing males of Prochyliza xanthostoma is intimidated, they fight, each striking the other with his head and antennae. Such fights may last for several minutes, a long conflict for such small, active, and delicate animals.
A male courts a female by dancing side-to-side, forequarters held high, displaying his elongated antennae and vibrating his elongated forelegs. A receptive female responds by spreading her forelegs to contact the male's long forelegs. The male somersaults over her forequarters and flips over to land on her back and lock onto her genitals. After several minutes of copulation, the female ejects a large part of the semen and swallows it. Such behaviour seems to correlate with the success of fertile egg production. Given such elaborate behavioural and morphological adaptations, waltzing flies are of interest as a model system for studying of the evolution of sexual dimorphism.[10][11]
Females oviposit on carrion, and larvae develop largely in sheltered parts of the carcass; depending on the degree of decomposition, they often develop inside the bone marrow. As in other piophilid species, final-instar larvae leap off the surface of the carcass where they had fed, and pupate in the soil.[12]
Medical and forensic significance
If swallowed (whether accidentally or otherwise), the larvae sometimes survive in the intestines and pass through the digestive system alive. Such behaviour is known as enteric or intestinal myiasis. In the gut, the larvae may cause serious lesions by attempting to bore through the intestinal walls. Symptoms include nausea, vomiting, pain in the abdomen, and bloody diarrhea. Both living and dead larvae may pass in the stool.[13] Some species also have been known to cause naso-oral and urogenital myiasis.[1]
In forensic entomology, the presence of P. casei larvae may be useful in estimating the date of death for human remains because they do not take up residence in a corpse until three to six months after death.[14] However, P. casei is not the only piophilid species to attack human corpses, so caution is appropriate in identification of the species found and in interpretation of their significance.[1]
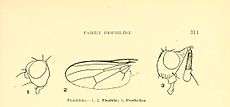
The family differs from the closely related family Sepsidae in several characters, particularly in having the costa broken at the end of the subcosta, the setulose mesonotum and the absence of a hair or fine bristle arising on the posterior edge of the posterior spiracle of the thorax.
Classification
The Piophilidae are a small family of less than 100 described species in 23 genera, mainly Holarctic in distribution, though some species are cosmopolitan. The nomenclature is volatile, with two subfamily names (Neottiophilinae and Thyreophorinae) in use recently, having been subsumed in the subfamily Piophilinae.
Recent works containing keys for identification of the Piophilidae include:
- McAlpine JF. 1977. A revised classification of the Piophilidae, including 'Neottiophilidae' and 'Thyreophoridae' (Diptera: Schizophora). Memoirs of the Entomological Society of Canada 103: 1-66
- McAlpine JF. (ed.) 1989. Manual of Nearctic Diptera (Vol. 3). Agriculture Canada Monograph No. 32.
- Ozerov, 2000. Piophilidae. In: Papp, L. & Darvas, A. (eds). Contributions to a Manual of Palaearctic Diptera. Appendix Volume. Science Herald, Budapest.pp 355–365. ISBN 963-04-8840-X
- Shtakel'berg, A.A. Family Piophilidae in Bei-Bienko, G. Ya, 1988 Keys to the insects of the European Part of the USSR Volume 5 (Diptera) Part 2 English edition
- Rochefort, S. et. al. (2015) Key to forensically important Piophilidae (Diptera) in the Nearctic Region. Canadian Journal of Arthropod Identification, 27: 1-37
A still useful older work is Séguy, E. (1934) Diptères: Brachycères. II. Muscidae acalypterae, Scatophagidae. Paris: Éditions Faune de France 28 BibliothequeVirtuelleNumerique pdf
See also
Genera
Species lists
Image galleries
References
- 1 2 3 Dorothy Gennard (16 March 2012). Forensic Entomology: An Introduction. John Wiley & Sons. pp. 67–. ISBN 978-1-119-94543-7. Retrieved 13 April 2013.
- ↑ Gary R. Mullen; Lance A. Durden (27 September 2002). Medical and Veterinary Entomology. Academic Press. pp. 139–. ISBN 978-0-08-053607-1. Retrieved 13 April 2013.
- ↑ G. Gordh; David H. Headrick (2001). A dictionary of entomology. CABI. pp. 701–. ISBN 978-0-85199-291-4. Retrieved 13 April 2013.
- 1 2 Don C. Mote (1914). "The cheese skipper (Piophila casei Linne)" (PDF). The Ohio Naturalist. 14 (7): 309–315.
- ↑ Maitland, David P. Locomotion by jumping in the Mediterranean fruit-fly larva Ceratitis capitata. Letters to Nature, Nature 355, 159 - 161 (9 January 1992); doi:10.1038/355159a0
- ↑ Antler fly (University of New South Wales)
- ↑ Bonduriansky, R. & Brooks, R.J. Why do male antler flies (Protopiophila litigata) fight? Ethology Ecology & Evolution 11: 287-301, 1999
- ↑ Bonduriansky, R.; Brassil, C.E. (2002). "Rapid and costly ageing in wild male flies". Nature. 420: 377. doi:10.1038/420377a.
- ↑ http://www.bonduriansky.net/waltzingflies.htm
- ↑ Bonduriansky, Russell, Wheeler, Jill, & Locke Rowe. Ejaculate feeding and female fitness in the sexually dimorphic fly Prochyliza xanthostoma (Diptera: Piophilidae). Animal Behaviour, 2005, 69, 489–497 doi:10.1016/j.anbehav.2004.03.018
- ↑ Bonduriansky, R. & Rowe, L. 2003. Interactions among mechanisms of sexual selection on male body size and head shape in a sexually dimorphic fly. Evolution, 57, 2046–2053.
- ↑ Bonduriansky, R. 2003. Layered sexual selection: a comparative analysis of sexual behaviour within an assemblage of piophilid flies. Canadian Journal of Zoology, 81, 479–491.
- ↑ David Alan Warrell; Timothy M. Cox; John D. Firth (2003). Oxford Textbook of Medicine: Sections 1-10. Oxford University Press. pp. 853–. ISBN 978-0-19-857014-1. Retrieved 11 April 2013.
- ↑ "Discovery Channel: You're on the Case". Archived from the original on 16 March 2006. Retrieved 14 November 2005.
- Berenbaum, May R. (1993). Ninety-Nine More Maggots, Mites, and Munchers. University of Illinois Press. ISBN 0-252-06322-8.
- Lieutenant Brian F. Prendergast, USN (2001). Filth Flies: Significance, Surveillance and Control in Contingency Operations (.pdf format). Retrieved 1 October 2005.
- Robinson, W H (2005). Handbook of Urban Insects And Arachnids: A Handbook of Urban Entomology. Cambridge University Press. ISBN 0-521-81253-4. pp. 180–181. Google Books
External links
 Data related to Piophilidae at Wikispecies
Data related to Piophilidae at Wikispecies- Family description and images
- An extensive bibliography on the Piophilidae
- Piophila casei on the UF / IFAS Featured Creatures Web site (note that many of the images on this site show other species misidentified as Piophila casei)
