Interstitial lung disease
| Interstitial lung disease | |
|---|---|
.jpg) | |
| End-stage pulmonary fibrosis of unknown origin, taken from an autopsy in the 1980s. | |
| Classification and external resources | |
| Specialty | Pulmonology |
| ICD-10 | J84.9 |
| ICD-9-CM | 518.89, 508.1, 515, 516.3, 714.81, 770.7 |
Interstitial lung disease (ILD), or diffuse parenchymal lung disease (DPLD),[1] is a group of lung diseases affecting the interstitium (the tissue and space around the air sacs of the lungs).[2] It concerns alveolar epithelium, pulmonary capillary endothelium, basement membrane, perivascular and perilymphatic tissues. It may occur when an injury to the lungs triggers an abnormal healing response. Ordinarily, the body generates just the right amount of tissue to repair damage. But in interstitial lung disease, the repair process goes awry and the tissue around the air sacs (alveoli) becomes scarred and thickened. This makes it more difficult for oxygen to pass into the bloodstream. The term ILD is used to distinguish these diseases from obstructive airways diseases.
Prolonged ILD may result in pulmonary fibrosis, but this is not always the case. Idiopathic pulmonary fibrosis is interstitial lung disease for which no obvious cause can be identified (idiopathic), and is associated with typical radiographic (basal and pleural based fibrosis with honeycombing) and pathologic (temporally and spatially heterogeneous fibrosis, histopathologic honeycombing and fibroblastic foci) findings.
In 2013 interstitial lung disease affected 595,000 people globally.[3] This resulted in 471,000 deaths.[4]
Causes
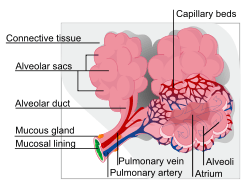
ILD may be classified according to the cause.[5] One method of classification is as follows:
- Inhaled substances
- Inorganic
- Organic
- Drug-induced
- Connective tissue and Autoimmune diseases
- Infection
- Idiopathic
- Malignancy
Diagnosis
Investigation is tailored towards the symptoms and signs. A proper and detailed history looking for the occupational exposures, and for signs of conditions listed above is the first and probably the most important part of the workup in patients with interstitial lung disease. Pulmonary function tests usually show a restrictive defect with decreased diffusion capacity (DLCO).
A lung biopsy is required if the clinical history and imaging are not clearly suggestive of a specific diagnosis or malignancy cannot otherwise be ruled out. In cases where a lung biopsy is indicated, a trans-bronchial biopsy is usually unhelpful, and a surgical lung biopsy is often required.
X-rays
Chest radiography is usually the first test to detect interstitial lung diseases, but the chest radiograph can be normal in up to 10% of patients, especially early on the disease process.[6][7]
High resolution CT of the chest is the preferred modality, and differs from routine CT of the chest. Conventional (regular) CT chest examines 7–10 mm slices obtained at 10 mm intervals; high resolution CT examines 1-1.5 mm slices at 10 mm intervals using a high spatial frequency reconstruction algorithm. The HRCT therefore provides approximately 10 times more resolution than the conventional CT chest, allowing the HRCT to elicit details that cannot otherwise be visualized.[6]
Radiologic appearance alone however is not adequate and should be interpreted in the clinical context, keeping in mind the temporal profile of the disease process.[6]
Interstitial lung diseases can be classified according to radiologic patterns.[6]
Pattern of opacities
- Consolidation
Acute: Alveolar hemorrhage syndromes, acute eosinophilic pneumonia, acute interstitial pneumonia, cryptogenic organizing pneumonia
Chronic: Chronic eosinophilic pneumonia, cryptogenic organizing pneumonia, lymphoproliferative disorders, pulmonary alveolar proteinosis, sarcoidosis
- Linear or reticular opacities
Acute: Pulmonary edema
Chronic: Idiopathic pulmonary fibrosis, connective tissue associated interstitial lung diseases, asbestosis, sarcoidosis, hypersensitivity pneumonitis, drug-induced lung disease
- Small nodules
Acute: Hypersensitivity pneumonitis
Chronic: Hypersensitivity pneumonitis, sarcoidosis, silicosis, coal workers pneumoconiosis, respiratory bronchiolitis, alveolar microlithiasis
- Cystic airspaces
Chronic: Pulmonary langerhans cell histiocytosis, pulmonary lymphangioleiomyomatosis, honeycomb lung caused by IPF or other diseases
Ground glass opacities
Acute: Alveolar hemorrhage syndromes, pulmonary edema, hypersensitivity pneumonitis, acute inhalational exposures, drug-induced lung diseases, acute interstitial pneumonia
Chronic: Nonspecific interstitial pneumonia, respiratory bronchiolitis associated interstitial lung disease, desquamative interstitial pneumonia, drug-induced lung diseases, pulmonary alveolar proteinosis
- Thickened alveolar septa
Acute: Pulmonary edema
Chronic: Lymphangitic carcinomatosis, pulmonary alveolar proteinosis, sarcoidosis, pulmonary veno occlusive disease[6]
Distribution
- Upper lung predominance
Pulmonary Langerhans cell histiocytosis, silicosis, coal workers pneumoconiosis, carmustine related pulmonary fibrosis, respiratory broncholitis associated with interstitial lung disease.
- Lower lung predominance
Idiopathic pulmonary fibrosis, pulmonary fibrosis associated with connective tissue diseases, asbestosis, chronic aspiration
- Central predominance (perihilar)
Sarcoidosis, berylliosis
- Peripheral predominance
Idiopathic pulmonary fibrosis, chronic eosinophilic pneumonia, cryptogenic organizing pneumonia[6]
Associated findings
- Pleural effusion or thickening
Pulmonary edema, connective tissue diseases, asbestosis, lymphangitic carcinomatosis, lymphoma, lymphangioleiomyomatosis, drug-induced lung diseases
- Lymphadenopathy
Sarcoidosis, silicosis, berylliosis, lymphangitic carcinomatosis, lymphoma, lymphocytic interstitial pneumonia[6]
Treatment
ILD is not a single disease, but encompasses many different pathological processes. Hence treatment is different for each disease.
If a specific occupational exposure cause is found, the person should avoid that environment. If a drug cause is suspected, that drug should be discontinued.
Many cases due to unknown or connective tissue-based causes are treated with corticosteroids,[8] such as prednisolone. Some people respond to immunosuppressant treatment. Patients with a low level of oxygen in the blood may be given supplemental oxygen.
Pulmonary rehabilitation appears to be useful.[9] Lung transplantation is an option if the ILD progresses despite therapy in appropriately selected patients with no other contraindications.[10][11]
On October 16, 2014, the Food and Drug Administration approved a new drug for the treatment of Idiopathic Pulmonary Fibrosis (IPF). This drug, Ofev (nintedanib), is marketed by Boehringer Ingelheim Pharmaceuticals, Inc. This drug has been shown to slow the decline of lung function although the drug has not been shown to reduce mortality or improve lung function. The estimated cost of the drug per year is approximately $94,000.[12]
References
- ↑ King TE (August 2005). "Clinical advances in the diagnosis and therapy of the interstitial lung diseases". Am. J. Respir. Crit. Care Med. 172 (3): 268–79. doi:10.1164/rccm.200503-483OE. PMID 15879420.
- ↑ "Frequently Asked Questions About Interstitial Lung Disease - University of Chicago Medical Center".
- ↑ Global Burden of Disease Study 2013, Collaborators (22 August 2015). "Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013.". Lancet (London, England). 386 (9995): 743–800. doi:10.1016/s0140-6736(15)60692-4. PMID 26063472.
- ↑ GBD 2013 Mortality and Causes of Death, Collaborators (10 January 2015). "Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013.". Lancet (London, England). 385 (9963): 117–71. doi:10.1016/s0140-6736(14)61682-2. PMC 4340604
 . PMID 25530442.
. PMID 25530442. - ↑ Bourke SJ (August 2006). "Interstitial lung disease: progress and problems". Postgrad Med J. 82 (970): 494–9. doi:10.1136/pgmj.2006.046417. PMC 2585700
 . PMID 16891438.
. PMID 16891438. - 1 2 3 4 5 6 7 Ryu, JH; Olson, EJ; Midthun, DE; Swensen, SJ (Nov 2002). "Diagnostic approach to the patient with diffuse lung disease.". Mayo Clinic Proceedings. 77 (11): 1221–7; quiz 1227. doi:10.4065/77.11.1221. PMID 12440558.
- ↑ "Interstitial Lung Disease/Pulmonary Fibrosis Northwestern Chicago Frequently Asked Questions".
- ↑ "Interstitial lung disease: Treatments and drugs - MayoClinic.com".
- ↑ Dowman, L; Hill, CJ; Holland, AE (Oct 6, 2014). "Pulmonary rehabilitation for interstitial lung disease.". The Cochrane database of systematic reviews. 10: CD006322. doi:10.1002/14651858.CD006322.pub3. PMID 25284270.
- ↑ Kotloff, RM; Thabut, G (Jul 15, 2011). "Lung transplantation.". American Journal of Respiratory and Critical Care Medicine. 184 (2): 159–71. doi:10.1164/rccm.201101-0134CI. PMID 21471083.
- ↑ Whelan, TP (Mar 2012). "Lung transplantation for interstitial lung disease.". Clinics in chest medicine. 33 (1): 179–89. doi:10.1016/j.ccm.2011.12.003. PMID 22365254.
- ↑ http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm418994.htm
