Incontinentia pigmenti
| Incontinentia pigmenti | |
|---|---|
| Classification and external resources | |
| Specialty | medical genetics |
| ICD-10 | Q82.3 |
| ICD-9-CM | 757.33 |
| OMIM | 308300 |
| DiseasesDB | 29600 |
| MedlinePlus | 001583 |
| eMedicine | article/1114205 article/1176285 |
| MeSH | D007184 |
| GeneReviews | |
| Orphanet | 464 |
Incontinentia pigmenti (IP, also known as "Bloch–Siemens syndrome,"[1] "Bloch–Sulzberger disease,"[2] "Bloch–Sulzberger syndrome"[1] "melanoblastosis cutis," and "nevus pigmentosus systematicus") is a genetic disorder that affects the skin, hair, teeth, nails, and central nervous system. It is named due to its microscopic appearance.
Presentation
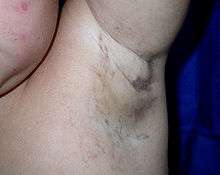
The skin lesions evolve through characteristic stages:
- blistering (from birth to about four months of age),
- a wart-like rash (for several months),
- swirling macular hyperpigmentation (from about six months of age into adulthood), followed by
- linear hypopigmentation.
Alopecia, hypodontia, abnormal tooth shape, and dystrophic nails are observed. Some patients have retinal vascular abnormalities predisposing to retinal detachment in early childhood. Cognitive delays/mental retardation are occasionally seen.
Discolored skin is caused by excessive deposits of melanin (normal skin pigment). Most newborns with IP will develop discolored skin within the first two weeks. The pigmentation involves the trunk and extremities, is slate-grey, blue or brown, and is distributed in irregular marbled or wavy lines. The discoloration sometimes fades with age.
Neurological problems can include: cerebral atrophy, the formation of small cavities in the central white matter of the brain, and the loss of neurons in the cerebellar cortex. About 20% of children with IP will have slow motor development, muscle weakness in one or both sides of the body, mental retardation, and seizures. They are also likely to have visual problems, which can include: crossed eyes, cataracts, and severe visual loss. Dental problems are common, and include missing or peg-shaped teeth - patients with IP often keep milk teeth into adult life.
Breast anomalies can occur in 1% of patients; anomalies can include hypoplasia and supernumerary nipples.
Skeletal and structural anomalies can occur in approximately 14% of patients, including:
- Somatic asymmetry,
- Hemivertebrae,
- Scoliosis,
- Spina bifida,
- Syndactyly,
- Acheiria (congenital absence of the hands - note: other limbs may be affected),
- Ear anomalies,
- Extra ribs,
- Skull deformities,
- Primary pulmonary hypertension,
- Cardiopulmonary failure
Genetics
IP is inherited in an X-linked dominant manner.[3][4] IP is lethal in most, but not all, males. A female with IP may have inherited the IKBKG mutation from either parent or have a new gene mutation. Parents may either be clinically affected or have germline mosaicism. Affected women have a 50% risk of transmitting the mutant IKBKG allele at conception; however, most affected male conceptuses miscarry. Thus, the expected ratio for liveborn children is 33% unaffected females, 33% affected females, and 33% unaffected males. Genetic counseling, prenatal testing, and preimplantation genetic diagnosis is available.
In females, the cells expressing the mutated IKBKG gene due to lyonization selectively die around the time of birth so the X-inactivation is extremely skewed.[5]
IP is caused by mutations in a gene called NEMO (NF-κB essential modulator).
Diagnosis
The diagnosis of IP is established by clinical findings and occasionally by corroborative skin biopsy. Molecular genetic testing of the NEMO IKBKG gene (chromosomal locus Xq28) reveals disease-causing mutations in about 80% of probands. Such testing is available clinically.
In addition, females with IP have skewed X-chromosome inactivation; testing for this can be used to support the diagnosis.
Many people in the past were misdiagnosed with a second type of IP, formerly known as IP1. This has now been given its own name - 'Hypomelanosis of Ito' (incontinentia pigmenti achromians). This has a slightly different presentation: swirls or streaks of hypopigmentation and depigmentation. It is not inherited and does not involve skin stages 1 or 2. Some 33–50% of patients have multisystem involvement — eye, skeletal, and neurological abnormalities. Its chromosomal locus is at Xp11, rather than Xq28.
History
This disorder was first reported by Bruno Bloch, a German dermatologist in 1926 and Marion Sulzberger, an American dermatologist in 1928.[6][7][8]
See also
- List of cutaneous conditions
- List of radiographic findings associated with cutaneous conditions
- List of dental abnormalities associated with cutaneous conditions
References
- 1 2 Rapini, Ronald P.; Bolognia, Jean L.; Jorizzo, Joseph L. (2007). Dermatology: 2-Volume Set. St. Louis: Mosby. ISBN 1-4160-2999-0.
- ↑ M. B. Sulzberger (1927). Über eine bisher nicht beschriebene kongenitale Pigmentanomalie (incontinentia pigmenti).. Berlin: Archiv für Dermatologie und Syphilis. p. 154.
- ↑ Pettigrew R, Kuo HC, Scriven P, et al. (December 2000). "A pregnancy following PGD for X-linked dominant [correction of X-linked autosomal dominant] incontinentia pigmenti (Bloch-Sulzberger syndrome): case report". Hum. Reprod. 15 (12): 2650–2. doi:10.1093/humrep/15.12.2650. PMID 11098039.
- ↑ "Incontinentia pigmenti. DermNet NZ".
- ↑ Smahi A, Courtois G, Vabres P, et al. (2000). "Genomic rearrangement in NEMO impairs NF-kappaB activation and is a cause of incontinentia pigmenti. The International Incontinentia Pigmenti (IP) Consortium". Nature. 405 (6785): 466–72. doi:10.1038/35013114. PMID 10839543.
- ↑ Bloch-Sulzberger pigment dermatosis (Bruno Bloch) at Who Named It?
- ↑ Bloch, B. (1926). "Eigentümliche, bisher nicht beschriebene Pigmentaffektion (incontinentia pigmenti)". Schweizerische medizinische Wochenschrift. Basel. 56: 404–5.
- ↑ Sulzberger, M.B. (1927). "Über eine bisher nicht beschriebene kongenitale Pigmentanomalie (incontinentia pigmenti)". Archiv für Dermatologie und Syphilis. Berlin. 154: 19–32. doi:10.1007/bf01828398.
External links
- Incontinentia Pigmenti
- Incontinentia pigmenti France
- Incontinentia Pigmenti Video
- GeneReview/NIH/UW entry on Incontinentia Pigmenti
- DermAtlas 166