Adenomyosis
| Adenomyosis | |
|---|---|
|
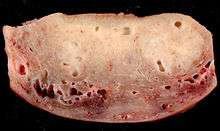
Adenomyosis uteri seen during laparoscopy: soft and enlarged uterus; the blue spots represent subserous endometriosis. | |
| Classification and external resources | |
| Specialty | Gynecology |
| ICD-10 | N80.0 |
| ICD-9-CM | 617.0 |
| OMIM | 600458 |
| DiseasesDB | 250 |
| MedlinePlus | 001513 |
| eMedicine | radio/737 |
| MeSH | D004715 |
Adenomyosis is a medical condition characterized by the abnormal presence endometrial tissue (the inner lining of the uterus) within the myometrium (the thick, muscular wall of the uterus).[1] Previously named as endometriosis interna, but adenomyosis differs from endometriosis and these two diseases represent two separate entities. They are found together in many cases.[2][3]
The term "adenomyometritis" specifically implies involvement of the uterus.[4][5]
The condition is typically found in women between the ages of 35 and 50 but can also be present in younger women.[6] Patients with adenomyosis often present with painful and/or profuse menses (dysmenorrhea & menorrhagia, respectively). Other possible symptoms are pain during sexual intercourse, chronic pelvic pain and irritation of the urinary bladder.
In adenomyosis, basal endometrium penetrates into hyperplastic myometrial fibers. Therefore, unlike functional layer, basal layer does not undergo typical cyclic changes with menstrual cycle.[3][7]
Adenomyosis may involve the uterus focally, creating an adenomyoma. With diffuse involvement, the uterus becomes bulky and heavier.
Signs and symptoms
Some women with adenomyosis do not experience any symptoms (30%), while others may have severe, debilitating symptoms.
Other symptoms include;[8]
- Intense debilitating abdominal or pelvic pain
- Strong 'contraction' feel of uterus
- Abdominal cramps
- A 'bearing' down feeling
- Pressure on bladder
- Dragging sensation down thighs and legs
- Heavy bleeding and flooding
- Large blood clots
Fertility
Adenomyosis itself can cause infertility issues, however chances for fertility can be improved if the adenomyosis has resolved following hormone therapies like levonorgestrel therapy. The discontinuation of medication or removal of IUD can be timed to be coordinated with fertility treatments. There has also been one report of successful pregnancy and healthy birth following high frequency ultrasound ablation of adenomyosis.
Complications
Adenomyosis is associated with an increased incidence of preterm labour and premature rupture of membranes.[9] Women with adenomyosis are at an increased risk of anemia due to increased blood loss during menses. This can cause fatigue, dizziness, and moodiness.
A review in 2012 found no evidence that adenomyosis should be detected and treated in patients who seek assisted reproduction treatment (ART), while other authors suggest checking every patient retrieving ART.[9]
Causes
The cause of adenomyosis is unknown, although it has been associated with any sort of uterine trauma that may break the barrier between the endometrium and myometrium, known as the junctional zone, such as a caesarean section, surgical pregnancy termination, and any pregnancy. It can be linked with endometriosis,[10] but studies looking into similarities and differences between these two conditions have conflicting results.[11]
The pathogenesis of adenomyosis still remains unclear, but the functioning of the inner myometrium, also called junction zone (JZ), is believed to play a major role in the development of adenomyosis. It is also a matter of discussion if the link between reproductive disorders and major obstetrical disorders also lies here.[12] Parity, age and previous uterine abrasion increase the risk of adenomyosis. Hormonal factors such as local hyperestrogenism and elevated levels of s-prolactin as well as autoimmune factors have also been identified as possible risk factors.[13][14][15] As both the myometrium and stroma in an adenomyosis affected uterus show significant differences from those of a non-affected uterus, a complex origin that includes multifactorial changes on both genetic and biochemical levels is likely.[16][17]
The tissue injury and repair (TIAR) theory is now widely accepted and suggests that uterine hyperperistalsis (i.e., increased peristalsis), during early periods of reproductive life will induce micro-injury at the endometrial-myometrial interface (EMI) region.[18] That again leads to elevation of local estrogen in order to heal the damage. At the same time, estrogen treatment will increase uterine peristalsis again, leading to a vicious circle and a chain of biological alterations essential for the development of adenomyosis. Iatrogenic injury of the junctional zone or physiological damages due to placental implantation most likely results in the same pathological cascade.[19] This also explains that adenomyosis often gets more severe after each pregnancy and childbirth, while endometriosis will be better.
Diagnosis
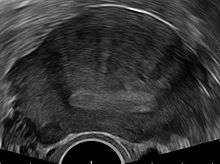
The uterus may be imaged using transvaginal ultrasound (US) or magnetic resonance imaging (MRI). Transvaginal ultrasound is the most cost effective and most available method. The diagnostical features are:[20][21]
- a globular enlarged uterus
- anterior/posterior wall asymmetry
- myometrial cysts
- linear, acoustic shadowing without presence of myoma
- diffuse myometrial echogenicity
- sign of endometrial invasion to the myometria
The junction zone (JZ) may be assessed by three-dimensional transvaginal ultrasound (3D TVUS) and features of adenomyosis are disruption, enlargement or invasion of the junctional zone.[22]
MRI provides better diagnostic capability due to the increased soft tissue differentiation, allowable through higher spatial and contrast resolution. MRI is limited by other factors, but not by calcified uterine fibroids (as is ultrasound). In particular, MRI is better able to differentiate adenomyosis from multiple small uterine fibroids. The uterus will have a thickened junctional zone with diminished signal on both T1 and T2 weighted sequences due to susceptibility effects of iron deposition due to chronic microhemorrhage. A thickness of the junctional zone greater than 10 to 12 mm (depending on who you read) is diagnostic of adenomyosis (<8 mm is normal). Interspersed within the thickened, hypointense signal of the junctional zone, one will often see foci of hyperintensity (brightness) on the T2 weighted scans representing small cystically dilatated glands or more acute sites of microhemorrhage.
MRI can be used to classify adenomyosis based on the depth of penetration of the ectopic endometrium into the myometrium.
Exact diagnosis of adenomyosis only possible in hysterectomy specimen.[3]
Differential diagnosis
The differential of abnormal uterine bleeding includes
- endometrial polyps
- submucosal fibroids
- endometrial hyperplasia
- endometrial carcinoma
Treatment
Removal of the uterus (hysterectomy) is the most effective treatment of women suffering from adenomyosis and the only causal therapy.
Levonogestrel-releasing intrauterine devices show positive results in women suffering from adenomysosis.[23] Gonadotropin releasing hormone (GnRH)-analogues, Danazol, uterine embolization and endometrial ablation have been tried in order to relieve adnomyosis related symptoms and show some effect, but the studies are few in number, mainly with a retrospective study design and have small sample sizes.[24] Long-time use of GnRH-analogues is often associated with heavy side effects, loss of bone densitiy and increased risk of cardiovascular events, and therefore not feasible for young women. Furthermore, all present treatment options are irrelevant options for women trying to conceive. Exogenous progestogenic treatments have been found to be ineffective.[16] In IVF-settings long down-regulation prior to IVF might have a positive effect on pregnancy rates.[25]
Conservative symptomatic treatment often consists of anti-inflammatory medications, such as ibuprofen or other NSAIDs.
Surgical options may include endometrial ablation, laparoscopic myometrial electrocoagulation and adenomyoma excision. These have demonstrated positive results in several studies, though long-term data is lacking. A non-surgical procedure, uterine artery embolization may also be used to block the blood supply to the uterus. High frequency ultrasound surgical ablation is also being explored as a treatment for both focal and diffuse forms of adenomyosis over complete hysterectomy. Hysterectomy may be warranted in some cases where fertility is not desired, and all other treatments have failed.[26]
Prognosis
Adenomyosis is a benign but often progressing condition. It is advocated that adenomyosis poses no increased risk for cancer development. However, both entities could coexist and the endometrial tissue within the myometrium could harbor endometrial adenocarcinoma, with potentially deep myometrial invasion.[27] As the condition is estrogen-dependent, menopause presents a natural cure. Ultrasound features of adenomyosis will still be present after menopause. Patients with adenomyosis often also have leiomyomata and/or endometriosis.
Etymology
The term adenomyosis is derived from the Greek terms adeno- (meaning gland), myo- (meaning muscle), and -osis (meaning condition).
See also
References
- ↑ "adenomyosis" at Dorland's Medical Dictionary
- ↑ Lazzeri L, Di Giovanni A, Exacoustos C et al. Preoperative and postoperative clinical and transvaginal ultrasound findings of adenomyosis in patients with deep infiltrating endometriosis. Reprod. Sci. doi:10.1177/1933719114522520 (2014) (Epub ahead of print).
- 1 2 3 Katz VL (2007). Comprehensive gynecology (5th ed.). Philadelphia PA: Mosby Elsevier.
- ↑ "adenomyometritis" at Dorland's Medical Dictionary
- ↑ Matalliotakis, I.; Kourtis, A.; Panidis, D. (2003). "Adenomyosis". Obstetrics and gynecology clinics of North America. 30 (1): 63–82, viii. doi:10.1016/S0889-8545(02)00053-0. PMID 12699258.
- ↑ Brosens, I., et al., Uterine Cystic Adenomyosis: A Disease of Younger Women. J Pediatr Adolesc Gynecol, 2014.
- ↑ Leyendecker G, Herbertz M, Kunz G, Mall G. Endometriosis results from the dislocation of basal endometrium. Hum. Reprod. 17, 2725–2736 (2002).
- ↑ Shrestha, A., et al., Adenomyosis at hysterectomy: prevalence, patient characteristics, clinical profile and histopatholgical findings. Kathmandu Univ Med J (KUMJ), 2012. 10(37): p. 53-6.
- 1 2 Maheshwari, A.; Gurunath, S.; Fatima, F.; Bhattacharya, S. (2012). "Adenomyosis and subfertility: A systematic review of prevalence, diagnosis, treatment and fertility outcomes". Human Reproduction Update. 18 (4): 374–392. doi:10.1093/humupd/dms006.
- ↑ Leyendecker G, Kunz G, Kissler S, Wildt L (August 2006). "Adenomyosis and reproduction". Best Pract Res Clin Obstet Gynaecol. 20 (4): 523–46. doi:10.1016/j.bpobgyn.2006.01.008. PMID 16520094.
- ↑ Benagiano, G.; Brosens, I.; Habiba, M. (2013). "Structural and molecular features of the endomyometrium in endometriosis and adenomyosis". Human Reproduction Update. 20 (3): 386–402. doi:10.1093/humupd/dmt052. ISSN 1355-4786. PMID 24140719.
- ↑ Brosens, I., et al., The enigmatic uterine junctional zone: the missing link between reproductive disorders and major obstetrical disorders? Hum Reprod, 2010. 25(3): p. 569-74.
- ↑ Kitawaki, J., Adenomyosis: the pathophysiology of an oestrogen-dependent disease. Best Pract Res Clin Obstet Gynaecol, 2006. 20(4): p. 493-502.
- ↑ Kitawaki, J., et al., Oestrogen receptor-alpha gene polymorphism is associated with endometriosis, adenomyosis and leiomyomata. Hum Reprod, 2001. 16(1): p. 51-55.
- ↑ Ota, H., et al., Is adenomyosis an immune disease? Hum Reprod Update, 1998. 4(4): p. 360-7.
- 1 2 Bergeron, C., F. Amant, and A. Ferenczy, Pathology and physiopathology of adenomyosis. Best Pract Res Clin Obstet Gynaecol, 2006. 20(4): p. 511-21.
- ↑ Nepomnyashchikh, L.M., et al., Immunocytochemical analysis of proliferative activity of endometrial and myometrial cell populations in focal and stromal adenomyosis. Bull Exp Biol Med, 2013. 155(4): p. 512-7.
- ↑ Leyendecker, G., L. Wildt, and G. Mall, The pathophysiology of endometriosis and adenomyosis: tissue injury and repair. Arch Gynecol Obstet, 2009. 280(4): p. 529-38.
- ↑ Leyendecker, G., et al., Adenomyosis and endometriosis. Re-visiting their association and further insights into the mechanisms of auto-traumatisation. An MRI study. Arch Gynecol Obstet, 2014.
- ↑ Kepkep K, Tuncay YA, Göynümer G, Tutal E (2007). "Transvaginal sonography in the diagnosis of adenomyosis: which findings are most accurate?". Ultrasound Obstet Gynecol. 30 (3): 341–5. doi:10.1002/uog.3985. PMID 17659649.
- ↑ Sakhel, K. and A. Abuhamad, Sonography of adenomyosis. J Ultrasound Med, 2012. 31(5): p. 805-8.
- ↑ Exacoustos, C., et al., Adenomyosis: three-dimensional sonographic findings of the junctional zone and correlation with histology. Ultrasound Obstet Gynecol, 2011. 37(4): p. 471-9.
- ↑ Bragheto, A.M., et al., Effectiveness of the levonorgestrel-releasing intrauterine system in the treatment of adenomyosis diagnosed and monitored by magnetic resonance imaging. Contraception, 2007. 76(3): p. 195-9.
- ↑ Maheshwari, A., et al., Adenomyosis and subfertility: a systematic review of prevalence, diagnosis, treatment and fertility outcomes. Hum Reprod Update, 2012. 18(4): p. 374-92.
- ↑ Niu, Z., et al., Long-term pituitary downregulation before frozen embryo transfer could improve pregnancy outcomes in women with adenomyosis. Gynecol Endocrinol, 2013.
- ↑ , Levgur, M. (2007). "Therapeutic options for adenomyosis: a review". Archives of Gynecology and Obstetrics. 276 (1): 1–15. doi:10.1007/S00404-006-0299-8. PMID 17186255.
- ↑ Ismiil N, Rasty G, Ghorab Z, et al. (August 2007). "Adenomyosis involved by endometrial adenocarcinoma is a significant risk factor for deep myometrial invasion". Ann Diagn Pathol. 11 (4): 252–7. doi:10.1016/j.anndiagpath.2006.08.011. PMID 17630108.

