Periodic acid
| |||
| Orthoperiodic acid | |||
| |||
| Metaperiodic acid | |||
| Names | |||
|---|---|---|---|
Other names
| |||
| Identifiers | |||
| 10450-60-9 (orthoperiodic) 13444-71-8 (metaperiodic) | |||
| 3D model (Jmol) | (orthoperiodic): Interactive image (metaperiodic): Interactive image | ||
| ChemSpider | 23622 (orthoperiodic) 58684 (metaperiodic) | ||
| ECHA InfoCard | 100.030.839 | ||
| PubChem | 65185 (metaperiodic) | ||
| UNII | AK1D44L87G | ||
| |||
| |||
| Properties | |||
| H5IO6 (orthoperiodic) HIO4 (metaperiodic) | |||
| Molar mass | 227.941 g/mol (H5IO6) 190.91 g/mol (HIO4) | ||
| Appearance | Colourless crystals | ||
| Melting point | 128.5 °C (263.3 °F; 401.6 K)[1] | ||
| Solubility | soluble in water, alcohols | ||
| Hazards | |||
| Safety data sheet | External MSDS | ||
| EU classification (DSD) |
Oxidizer (O), Toxic (T), Corrosive (C) | ||
| R-phrases | R23 R24 R25 R34 R41 | ||
| NFPA 704 | |||
| Related compounds | |||
| Other anions |
| ||
| Other cations |
|||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
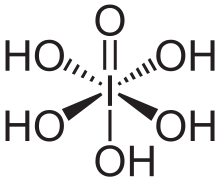
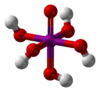
Periodic acid (pronounced per-iodic /ˌpɜːr.aɪˈɒdᵻk/ PURR-eye-OH-dik, rather than /ˌpɪərɪˈɒdᵻk/ PEER-ee-OD-ik as in periodic table) is the highest oxoacid of iodine, in which the iodine exists in oxidation state VII. Like all periodates it can exist in two forms: orthoperiodic acid, with the chemical formula H5IO6 and metaperiodic acid, which has the formula HIO4.
Periodic acid was discovered by Heinrich Gustav Magnus and C. F. Ammermüller in 1833.[2]
Synthesis
Modern industrial scale production involves the electrochemical oxidation of iodic acid, on a PbO2 anode, with the following standard electrode potential:
Orthoperiodic acid can be dehydrated to give metaperiodic acid by heating to 100 °C
- HIO4 + 2 H2O ⇌ H5IO6
Further heating to around 150 °C gives iodine pentoxide (I2O5) rather than the expected anhydride diiodine heptoxide (I2O7). Metaperiodic acid can also be prepared by from various orthoperiodates by treatment with dilute nitric acid.[4]
- H5IO6 → HIO4 + 2 H2O
Properties
Orthoperiodic acid has a number of acid dissociation constants.[5][6] The pKa of meta periodic acid has not been determined.
- H5IO6 ⇌ H
4IO−
6 + H+, pKa = 3.29
- H
4IO−
6 ⇌ H
3IO2−
6 + H+, pKa = 8.31
- H
3IO2−
6 ⇌ H
2IO3−
6 + H+, pKa = 11.60
There being two forms of periodic acid, it follows that two types of periodate salts are formed. For example, sodium metaperiodate, NaIO4, can be synthesised from HIO4 while sodium orthoperiodate, Na5IO6 can be synthesised from H5IO6.
Structure
Orthoperiodic acid forms monoclinic crystals (space group P21/n) consisting of slightly deformed IO6 octahedron interlinked via bridging hydrogens.[7] The crystal structure of metaperiodic acid also includes IO6 octahedron, however these are connected via cis-edge-sharing with bridging oxygens to form one-dimensional infinite chains.[8]
Reactions
Like all periodates periodic acid can be used to cleave various 1,2-difunctional compounds. Most notably periodic acid will cleave vicinal diols into two aldehyde or ketone fragments.
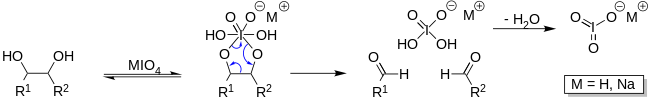
This can be useful in determining the structure of carbohydrates as periodic acid can be used to open saccharide rings. This process is often used in labeling saccharides with fluorescent molecules or other tags such as biotin. Because the process requires vicinal diols, periodate oxidation is often used to selectively label the 3′-termini of RNA (ribose has vicinal diols) instead of DNA as deoxyribose does not have vicinal diols.
Periodic acid is also used in as an oxidising agent of moderate strength.
Other oxyacids
Periodate is part of a series of oxyacids in which iodine can assume oxidation states of −1, +1, +3, +5, or +7. A number of neutral iodine oxides are also known.
| Iodine oxidation state | −1 | +1 | +3 | +5 | +7 |
|---|---|---|---|---|---|
| Name | Hydrogen iodide | Hypoiodous acid | Iodous acid | Iodic acid | Periodic acid |
| Formula | HI | HIO | HIO2 | HIO3 | HIO4 or H5IO6 |
See also
Compounds with a similar structure:
- Perchloric acid, perbromic acid and perastatic acid, the related perhalogenic acids
- Telluric acid and perxenic acid, the isoelectronic oxoacids of tellurium and xenon
Compounds with similar chemistry:
References
- ↑ Aylett, founded by A.F. Holleman ; continued by Egon Wiberg ; translated by Mary Eagleson, William Brewer ; revised by Bernhard J. (2001). Inorganic chemistry (1st English ed., [edited] by Nils Wiberg. ed.). San Diego, Calif. : Berlin: Academic Press, W. de Gruyter. p. 453. ISBN 0123526515.
- ↑ Ammermüller, F.; Magnus, G. (1833). "Ueber eine neue Verbindung des Jods mit Sauerstoff, die Ueberjodsäure". Annalen der Physik und Chemie (in German). 104 (7): 514–525. doi:10.1002/andp.18331040709.
- ↑ Parsons, Roger (1959). Handbook of electrochemical constants. Butterworths Scientific Publications Ltd. p. 71.
- ↑ Riley, edited by Georg Brauer; translated by Scripta Technica, Inc. Translation editor Reed F. (1963). Handbook of preparative inorganic chemistry. Volume 1 (2nd ed.). New York, N.Y.: Academic Press. pp. 323–324. ISBN 012126601X.
- ↑ Aylett, founded by A.F. Holleman; continued by Egon Wiberg; translated by Mary Eagleson, William Brewer; revised by Bernhard J. (2001). Inorganic chemistry (1st English ed., [edited] by Nils Wiberg. ed.). San Diego, Calif. : Berlin: Academic Press, W. de Gruyter. p. 454. ISBN 0123526515.
- ↑ Burgot, Jean-Louis. Ionic equilibria in analytical chemistry. New York: Springer. p. 358. ISBN 1441983821.
- ↑ Feikema, Y. D. (10 June 1966). "The crystal structures of two oxy-acids of iodine. I. A study of orthoperiodic acid, H5IO6, by neutron diffraction". Acta Crystallographica. 20 (6): 765–769. doi:10.1107/S0365110X66001828.
- ↑ Kraft, Thorsten; Jansen, Martin (1 September 1997). "Crystal Structure Determination of Metaperiodic Acid, HIO4, with Combined X-Ray and Neutron Diffraction". Angewandte Chemie International Edition in English. 36 (16): 1753–1754. doi:10.1002/anie.199717531.