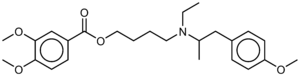
Mebeverine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Colofac, Duspamen and others |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | A03AA04 (WHO) |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number |
2753-45-9 |
| PubChem (CID) | 4031 |
| ChemSpider |
3891 |
| UNII |
7F80CC3NNV |
| KEGG |
D04868 |
| ChEMBL |
CHEMBL282121 |
| ECHA InfoCard | 100.018.546 |
| Chemical and physical data | |
| Formula | C25H35NO5 |
| Molar mass | 429.6 g/mol |
| 3D model (Jmol) | Interactive image |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Mebeverine is a drug whose major therapeutic role is in the treatment of irritable bowel syndrome (IBS) and the associated abdominal cramping. It works by relaxing the muscles in and around the gut. It is a musculotropic antispasmodic drug without anticholinergic side-effects. The drug is also indicated for treatment of gastrointestinal spasm secondary to organic disorder.
_135mg_UK.jpg)
Indications
Spastic functional disturbances of the colon:
- Irritable bowel syndrome in its primary form
- Irritable bowel syndrome associated with organic lesions of the gastrointestinal tract such as; diverticulosis and diverticulitis, regional enteritis, disease of the gall bladder and gall ducts, gastric and duodenal ulcers, dysentery, and aspecific or specific inflammation of the digestive tract.
- Mebeverine should be taken 20 minutes before meals.
Mechanism of action
Mebeverine is an antimuscarinic. It is also an inhibitor of calcium-depot replenishment. Musculotropic compounds act directly on the gut muscles at the cellular level to relax them. This relieves painful muscle spasms of the gut, without affecting its normal motility. Mebeverine is used to relieve symptoms of irritable bowel syndrome and related intestinal disorders that are the result of spasms in the intestinal muscles. These include colicky abdominal pain and cramps, diarrhoea alternating with constipation and flatulence (wind).
Adverse effects
Side effects may include:[1]
- Indigestion or heartburn
- Constipation
- Dizziness
- Insomnia (difficulty sleeping)
- anorexia (loss of appetite)
- Headache
- Tiredness
- Slow heart beat
- Rash and/or itchy skin
- Nausea
Since 1978, 21 cases of severe adverse reactions to mebeverine were reported in the Netherlands.[2] Most reactions consisted of urticaria or maculopapular rash, sometimes accompanied by fever, polyarthritis, thrombocytopenia or angioedema.
Very rarely, people taking this medicine may develop allergic reactions.[3]
Pregnancy and breastfeeding
Mebeverine passes into breast milk, but the amount is considered too small to be harmful to a nursing infant.[1]
Driving and using machines
Mebeverine is unlikely to affect the ability to operate machinery or to drive, yet not completely out of the question.[4][5]
History
It was first registered in 1965.[6]
Availability
Mebeverine is a generic drug and is available internationally under many brand names.[7]
References
- 1 2 Colofac Data Sheet (PDF) http://www.medsafe.govt.nz/profs/datasheet/c/colofactab.pdf
- ↑ in 't Veld BA, van Puyenbroek E, Stricker BH (1997). Hypersensitivity reactions to use of mebeverine. pp. 1392–5. Ned Tijdschr Geneeskd. 1997 July 12;141(28):1392-5. Hypersensitivity reactions to use of mebeverine pubmed: 9380201
- ↑ Colofac Patient leaflet sheet http://emc.medicines.org.uk/medicine/2531/PIL/Colofac+Tablets+135mg/
- ↑ http://www.drugs.com/uk/mebeverine-50mg-5ml-sugar-free-oral-suspension-leaflet.html
- ↑ http://www.nhs.uk/medicine-guides/pages/MedicineOverview.aspx?condition=Irritable%20bowel&medicine=mebeverine%20hydrochloride&preparation=Mebeverine%20135mg%20tablets
- ↑ Mebeverine page at druginfosys Page accessed Feb 1, 2015
- ↑ International page at drugs.com for Mebeverine Page accessed Feb 1, 2015