Tyrosine
 L-Tyrosine | |
 L-Tyrosine at physiological pH | |
| Names | |
|---|---|
| IUPAC name
(S)-Tyrosine | |
| Other names
L-2-Amino-3-(4-hydroxyphenyl)propanoic acid | |
| Identifiers | |
| 60-18-4 (L) | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:58315 |
| ChEMBL | ChEMBL925 |
| ChemSpider | 5833 |
| DrugBank | DB03839 |
| ECHA InfoCard | 100.000.419 |
| 4791 | |
| PubChem | 1153 |
| UNII | 42HK56048U |
| |
| |
| Properties | |
| C9H11NO3 | |
| Molar mass | 181.19 g·mol−1 |
| Hazards | |
| Safety data sheet | See: data page |
| NFPA 704 | |
| Supplementary data page | |
| Refractive index (n), Dielectric constant (εr), etc. | |
| Thermodynamic data |
Phase behaviour solid–liquid–gas |
| UV, IR, NMR, MS | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Tyrosine (Tyr or Y)[1] or 4-hydroxyphenylalanine is one of the 22 amino acids that are used by cells to synthesize proteins. It is a non-essential amino acid with a polar side group. Its codons are UAC and UAU. The word "tyrosine" is from the Greek tyros, meaning cheese, as it was first discovered in 1846 by German chemist Justus von Liebig in the protein casein from cheese.[2][3] It is called tyrosyl when referred to as a functional group or side chain. Tyrosine is a hydrophobic amino acid and is significantly less soluble in water than its precursor, phenylalanine, due to the thermodynamic favorability of the hydrogen bonding between the hydroxyl group of one molecule of tyrosine and the carboxyl group of another.
Functions
Aside from being a proteinogenic amino acid, tyrosine has a special role by virtue of the phenol functionality. It occurs in proteins that are part of signal transduction processes. It functions as a receiver of phosphate groups that are transferred by way of protein kinases (so-called receptor tyrosine kinases). Phosphorylation of the hydroxyl group changes the activity of the target protein.
A tyrosine residue also plays an important role in photosynthesis. In chloroplasts (photosystem II), it acts as an electron donor in the reduction of oxidized chlorophyll. In this process, it loses the hydrogen atom of its phenolic OH-group. This radical is subsequently reduced in the photosystem II by the four core manganese clusters.
Dietary requirements and sources
A recommended daily intake for phenylalanine and tyrosine is 25 mg per kilogram of body weight, or 11 mg per pound.[4] For a 70 kg person this is 1750 mg (phenylalanine + tyrosine).
Tyrosine, which can also be synthesized in the body from phenylalanine, is found in many high-protein food products such as chicken, turkey, fish, milk, yogurt, cottage cheese, cheese, peanuts, almonds, pumpkin seeds, sesame seeds, soy products, lima beans, avocados, and bananas.[5] For example, the white of an egg has about 250 mg per egg,[4] while lean beef/lamb/pork/salmon/chicken/turkey contains about 1000 mg per 3 ounces (85 g) portion.[4]
Biosynthesis

In plants and most microorganisms, tyr is produced via prephenate, an intermediate on the shikimate pathway. Prephenate is oxidatively decarboxylated with retention of the hydroxyl group to give p-hydroxyphenylpyruvate, which is transaminated using glutamate as the nitrogen source to give tyrosine and α-ketoglutarate.
Mammals synthesize tyrosine from the essential amino acid phenylalanine (phe), which is derived from food. The conversion of phe to tyr is catalyzed by the enzyme phenylalanine hydroxylase, a monooxygenase. This enzyme catalyzes the reaction causing the addition of a hydroxyl group to the end of the 6-carbon aromatic ring of phenylalanine, such that it becomes tyrosine.
Metabolism

Phosphorylation and sulfation
Some of the tyrosine residues can be tagged (at the hydroxyl group) with a phosphate group (phosphorylated) by protein kinases. In its phosphorylated form, tyrosine is called phosphotyrosine. Tyrosine phosphorylation is considered to be one of the key steps in signal transduction and regulation of enzymatic activity. Phosphotyrosine can be detected through specific antibodies. Tyrosine residues may also be modified by the addition of a sulfate group, a process known as tyrosine sulfation.[6] Tyrosine sulfation is catalyzed by tyrosylprotein sulfotransferase (TPST). Like the phosphotyrosine antibodies mentioned above, antibodies have recently been described that specifically detect sulfotyrosine.
Precursor to neurotransmitters and hormones
In dopaminergic cells in the brain, tyrosine is converted to L-DOPA by the enzyme tyrosine hydroxylase (TH). TH is the rate-limiting enzyme involved in the synthesis of the neurotransmitter dopamine. Dopamine can then be converted into catecholamines, such as norepinephrine (noradrenaline) and epinephrine (adrenaline).
The thyroid hormones triiodothyronine (T3) and thyroxine (T4) in the colloid of the thyroid also are derived from tyrosine.
Precursor to alkaloids
The latex of Papaver somniferum, the opium poppy, has been shown to convert tyrosine into the alkaloid morphine and the bio-synthetic pathway has been established from tyrosine to morphine by using Carbon-14 radio-labelled tyrosine to trace the in-vivo synthetic route.
Mescaline producing cactus bio-synthesize tyrosine into mescaline when injected with it.[10]
Precursor to natural phenols
Tyrosine ammonia lyase (TAL) is an enzyme in the natural phenols biosynthesis pathway. It transforms L-tyrosine into p-coumaric acid.
Precursor to pigments
Tyrosine is also the precursor to the pigment melanin.
Role in coenzyme Q10 synthesis
Tyrosine (or its precursor phenylalanine) is needed to synthesize the benzoquinone structure which forms part of coenzyme Q10.
Degradation

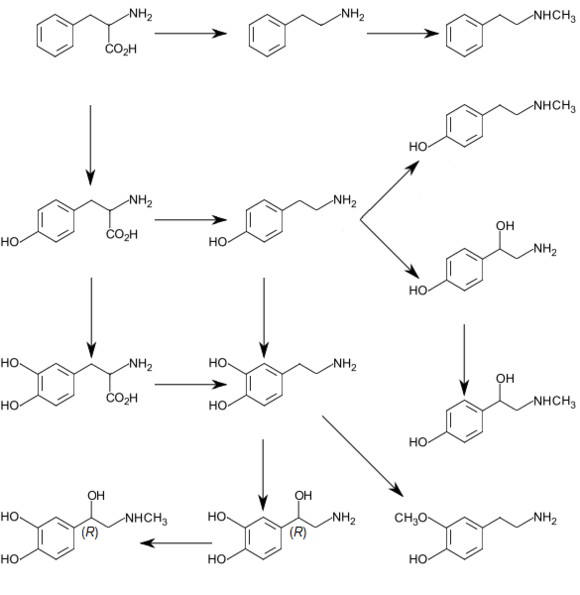
The decomposition of L-tyrosine (syn. para-hydroxyphenylalanine) begins with an α-ketoglutarate dependent transamination through the tyrosine transaminase to para-hydroxyphenylpyruvate. The positional description para, abbreviated p, mean that the hydroxyl group and side chain on the phenyl ring are across from each other (see the illustration below).
The next oxidation step catalyzes by p-hydroxylphenylpyruvate-dioxygenase and splitting off CO2 homogentisate (2,5-dihydroxyphenyl-1-acetate). In order to split the aromatic ring of homogentisate, a further dioxygenase, homogentistate-oxygenase is required. Thereby, through the incorporation of a further O2 molecule, maleylacetoacetate is created.
Fumarylacetate is created maleylacetoacetate-cis-trans-isomerase through rotation of the carboxyl group created from the hydroxyl group via oxidation. This cis-trans-isomerase contains glutathione as a coenzyme. Fumarylacetoacetate is finally split by the enzyme fumarylacetoacetate hydrolase through the addition of a water molecule.
Thereby fumarate (also a metabolite of the citric acid cycle) and acetoacetate (3-ketobutyroate) are liberated. Acetoacetate is a ketone body, which is activated with succinyl-CoA, and thereafter it can be converted into acetyl-CoA, which in turn can be oxidized by the citric acid cycle or be used for fatty acid synthesis.
Phloretic acid is also a urinary metabolite of tyrosine in rats.[11]
Ortho- and meta-tyrosine

Three structural isomers of L-tyrosine are known. In addition to the common amino acid L-tyrosine, which is the para isomer (para-tyr, p-tyr or 4-hydroxyphenylalanine), there are two additional regioisomers, namely meta-tyrosine (also known as 3-hydroxyphenylalanine, L-m-tyrosine, and m-tyr) and ortho-tyrosine (o-tyr or 2-hydroxyphenylalanine), that occur in nature. The m-tyr and o-tyr isomers, which are rare, arise through non-enzymatic free-radical hydroxylation of phenylalanine under conditions of oxidative stress.[12][13]
m-Tyrosine and analogues (rare in nature but available synthetically) have shown application in Parkinson's Disease, Alzheimer's disease and arthritis.[14]
Medical use
Tyrosine is a precursor to neurotransmitters and increases plasma neurotransmitter levels (particularly dopamine and norepinephrine),[15] but has little if any effect on mood in normal subjects.[16][17][18] The effect on mood is noted in humans subjected to stressful conditions (see below).
A number of studies have found tyrosine to be useful during conditions of stress, cold, fatigue,[19] prolonged work and sleep deprivation,[20][21] with reductions in stress hormone levels,[22] reductions in stress-induced weight loss seen in animal trials,[19] and improvements in cognitive and physical performance[17][23][24] seen in human trials.
Tyrosine does not seem to have any significant effect on cognitive or physical performance in normal circumstances,[25][26] but does help sustain working memory better during multitasking.[27]
Industrial synthesis
L-tyrosine and its derivatives (L-DOPA, melanin, phenylpropanoids, and others) are used in pharmaceuticals, dietary supplements, and food additives. Two methods were formerly used to manufacture L-tyrosine. The first involves the extraction of the desired amino acid from protein hydrolysates using a chemical approach. The second utilizes enzymatic synthesis from phenolics, pyruvate, and ammonia through the use of tyrosine phenol-lyase.[28] Advances in genetic engineering and the advent of industrial fermentation have shifted the synthesis of L-tyrosine to the use of engineered strains of E. coli.[29][30][31]
See also
- Albinism
- Alkaptonuria
- Betalain
- Iodinated tyrosine derivatives
- Tyramine
- Tyrosine sulfation
- Tyrosinemia
- Pauly reaction
References
- ↑ IUPAC-IUBMB Joint Commission on Biochemical Nomenclature (1983). "Nomenclature and Symbolism for Amino Acids and Peptides". Recommendations on Organic & Biochemical Nomenclature, Symbols & Terminology. Retrieved 2007-05-17.
- ↑ "Tyrosine". The Columbia Electronic Encyclopedia, 6th ed. Infoplease.com — Columbia University Press. 2007. Retrieved 2008-04-20.
- ↑ Douglas Harper (2001). "Tyrosine". Online Etymology Dictionary. Retrieved 2008-04-20.
- 1 2 3 Top 10 Foods Highest in Tyrosine
- ↑ "Tyrosine". University of Maryland Medical Center. Retrieved 2011-03-17.
- ↑ Hoffhines AJ, Damoc E, Bridges KG, Leary JA, Moore KL (2006). "Detection and purification of tyrosine-sulfated proteins using a novel anti-sulfotyrosine monoclonal antibody". J. Biol. Chem. 281 (49): 37877–87. doi:10.1074/jbc.M609398200. PMC 1764208
 . PMID 17046811.
. PMID 17046811. - ↑ Broadley KJ (March 2010). "The vascular effects of trace amines and amphetamines". Pharmacol. Ther. 125 (3): 363–375. doi:10.1016/j.pharmthera.2009.11.005. PMID 19948186.
- ↑ Lindemann L, Hoener MC (May 2005). "A renaissance in trace amines inspired by a novel GPCR family". Trends Pharmacol. Sci. 26 (5): 274–281. doi:10.1016/j.tips.2005.03.007. PMID 15860375.
- ↑ Wang X, Li J, Dong G, Yue J (February 2014). "The endogenous substrates of brain CYP2D". Eur. J. Pharmacol. 724: 211–218. doi:10.1016/j.ejphar.2013.12.025. PMID 24374199.
The highest level of brain CYP2D activity was found in the substantia nigra (Bromek et al., 2010). The in vitro and in vivo studies have shown the contribution of the alternative CYP2D-mediated dopamine synthesis to the concentration of this neurotransmitter although the classic biosynthetic route to dopamine from tyrosine is active. CYP2D6 protein level is approximately 40% lower in the frontal cortex, cerebellum, and hippocampus in PD patients, even when controlling for CYP2D6 genotype (Mann et al., 2012). ... Tyramine levels are especially high in the basal ganglia and limbic system, which are thought to be related to individual behavior and emotion (Yu et al., 2003c). Studies have demonstrated that dopamine is formed from p-tyramine as well as m-tyramine via tyramine 3-hydroxylation or 4-hydroxylation by rat CYP2D2, 2D4, and 2D18 as well as human CYP2D6. ... Both rat CYP2D and human CYP2D6 have a higher affinity for m-tyramine compared with p-tyramine for the generation of dopamine. Rat CYP2D isoforms (2D2/2D4/2D18) are less efficient than human CYP2D6 for the generation of dopamine from p-tyramine. The Km values of the CYP2D isoforms are as follows: CYP2D6 (87–121 μm) ≈ CYP2D2 ≈ CYP2D18 > CYP2D4 (256 μm) for m-tyramine and CYP2D4 (433 μm) > CYP2D2 ≈ CYP2D6 > CYP2D18 (688 μm) for p-tyramine (Bromek et al., 2010; Thompson et al., 2000).
- ↑ "Erowid Cacti Vault : Cactus Growers Guide". Erowid.org. 2008-03-08. Retrieved 2013-04-16.
- ↑ Booth, A N; Masri, M S; Robbins, D J; Emerson, O H; Jones, F T; Deeds, F (1960). "Urinary phenolic acid metabolities of tyrosine". Journal of Biological Chemistry. 235 (9): 2649–2652.
- ↑ Molnár GA, Wagner Z, Markó L, Kó Szegi T, Mohás M, Kocsis B, Matus Z, Wagner L, Tamaskó M, Mazák I, Laczy B, Nagy J, Wittmann I (2005). "Urinary ortho-tyrosine excretion in diabetes mellitus and renal failure: evidence for hydroxyl radical production". Kidney Int. 68 (5): 2281–7. doi:10.1111/j.1523-1755.2005.00687.x. PMID 16221230.
- ↑ Molnár GA, Nemes V, Biró Z, Ludány A, Wagner Z, Wittmann I (2005). "Accumulation of the hydroxyl free radical markers meta-, ortho-tyrosine and DOPA in cataractous lenses is accompanied by a lower protein and phenylalanine content of the water-soluble phase". Free Radic. Res. 39 (12): 1359–66. doi:10.1080/10715760500307107. PMID 16298866.
- ↑ Humphrey, Cara E.; Furegati, Markus; Laumen, Kurt; La Vecchia, Luigi; Leutert, Thomas; Müller-Hartwieg, J. Constanze D.; Vögtle, Markus (2007). "Optimized Synthesis of L-m-Tyrosine Suitable for Chemical Scale-Up". Organic Process Research & Development. 11: 1069–1075. doi:10.1021/op700093y.
- ↑ Rasmussen DD, Ishizuka B, Quigley ME, Yen SS (1983). "Effects of tyrosine and tryptophan ingestion on plasma catecholamine and 3,4-dihydroxyphenylacetic acid concentrations". J. Clin. Endocrinol. Metab. 57 (4): 760–3. doi:10.1210/jcem-57-4-760. PMID 6885965.
- ↑ Leathwood PD, Pollet P (1982). "Diet-induced mood changes in normal populations". Journal of Psychiatric Research. 17 (2): 147–54. doi:10.1016/0022-3956(82)90016-4. PMID 6764931.
- 1 2 Deijen JB, Orlebeke JF (1994). "Effect of tyrosine on cognitive function and blood pressure under stress". Brain Res. Bull. 33 (3): 319–23. doi:10.1016/0361-9230(94)90200-3. PMID 8293316.
- ↑ Lieberman HR, Corkin S, Spring BJ, Wurtman RJ, Growdon JH (1985). "The effects of dietary neurotransmitter precursors on human behavior". Am J Clin Nutr. 42 (2): 366–370. PMID 4025206.
- 1 2 Hao S, Avraham Y, Bonne O, Berry EM (2001). "Separation-induced body weight loss, impairment in alternation behavior, and autonomic tone: effects of tyrosine". Pharmacol. Biochem. Behav. 68 (2): 273–81. doi:10.1016/S0091-3057(00)00448-2. PMID 11267632.
- ↑ Magill RA, Waters WF, Bray GA, Volaufova J, Smith SR, Lieberman HR, McNevin N, Ryan DH (2003). "Effects of tyrosine, phentermine, caffeine D-amphetamine, and placebo on cognitive and motor performance deficits during sleep deprivation". Nutritional Neuroscience. 6 (4): 237–46. doi:10.1080/1028415031000120552. PMID 12887140.
- ↑ Neri DF, Wiegmann D, Stanny RR, Shappell SA, McCardie A, McKay DL (1995). "The effects of tyrosine on cognitive performance during extended wakefulness". Aviation, space, and environmental medicine. 66 (4): 313–9. PMID 7794222.
- ↑ Reinstein DK, Lehnert H, Wurtman RJ (1985). "Dietary tyrosine suppresses the rise in plasma corticosterone following acute stress in rats". Life Sci. 37 (23): 2157–63. doi:10.1016/0024-3205(85)90566-1. PMID 4068899.
- ↑ Deijen JB, Wientjes CJ, Vullinghs HF, Cloin PA, Langefeld JJ (1999). "Tyrosine improves cognitive performance and reduces blood pressure in cadets after one week of a combat training course". Brain Res. Bull. 48 (2): 203–9. doi:10.1016/S0361-9230(98)00163-4. PMID 10230711.
- ↑ Mahoney CR, Castellani J, Kramer FM, Young A, Lieberman HR (2007). "Tyrosine supplementation mitigates working memory decrements during cold exposure". Physiology and Behavior. IN PRESS (4): 575–82. doi:10.1016/j.physbeh.2007.05.003. PMID 17585971.
- ↑ Chinevere TD, Sawyer RD, Creer AR, Conlee RK, Parcell AC (2002). "Effects of L-tyrosine and carbohydrate ingestion on endurance exercise performance". J. Appl. Physiol. 93 (5): 1590–7. doi:10.1152/japplphysiol.00625.2001 (inactive 2015-01-09). PMID 12381742.
- ↑ Strüder HK, Hollmann W, Platen P, Donike M, Gotzmann A, Weber K (1998). "Influence of paroxetine, branched-chain amino acids and tyrosine on neuroendocrine system responses and fatigue in humans". Horm. Metab. Res. 30 (4): 188–94. doi:10.1055/s-2007-978864. PMID 9623632.
- ↑ Thomas JR, Lockwood PA, Singh A, Deuster PA (1999). "Tyrosine improves working memory in a multitasking environment". Pharmacol. Biochem. Behav. 64 (3): 495–500. doi:10.1016/S0091-3057(99)00094-5. PMID 10548261.
- ↑ Lutke-Eversloh, T., Santos, C.N.S. (2007) Perspectives of biotechnological production of L-tyrosine and its applications. Appl. Microbiol. Biotechnol. 77: 751-762. PMID 17968539
- ↑ Chavez-Bejar, M., J. Baez-Viveros, A. Martinez, F. Bolivar, G. Gosset. (2012) Biotechnological production of L-tyrosine and derived compounds. Process Biochemistry. 47: 1017-1026
- ↑ Lutke-Eversloh, T., Santos, C.N.S. (2007) Perspectives of biotechnological production of L-tyrosine and its applications. Appl. Microbiol. Biotechnol. 77: 751-762
- ↑ Chavez-Bejar, M., J. Baez-Viveros, A. Martinez, F. Bolivar, G. Gosset. (2012) Biotechnological production of L-tyrosine and derived compounds. Process Biochemistry. 47: 1017-1026
External links
- Tyrosine MS Spectrum
- Tyrosine metabolism
- Phenylalanine and tyrosine biosynthesis
- Phenylalanine, Tyrosine, and tryptophan biosynthesis
- Tyrosine in the ChemIDplus database